
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
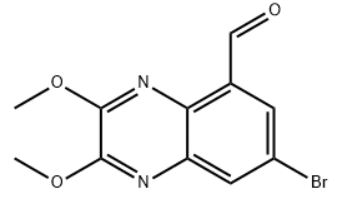
Product Name5-Quinoxalinecarboxaldehyde, 7-bromo-2,3-dimethoxy-IUPAC Name7-bromo-2,3-dimethoxyquinoxaline-5-carbaldehydeMolecular StructureCAS Registry Number 187479-66-9Synonyms7-Bromo-5-formyl-2,3-dimethoxy-quinoxaline187479-66-9SCHEMBL8078939VJEIGKGDNHGYNW-UHFFFAOYSA-NDB-341967Molecular FormulaC11H9BrN2O3Molecular Weight297.1InChIInChI=1S/C11H9BrN2O3/c1-16-10-11(17-2)14-9-6(5-15)3-7(12)4-8(9)13-10/h3-5H,1-2H3InChI KeyVJEIGKGDNHGYNW-UHFFFAOYSA-NSMILESCOC1=NC2=CC(=CC(=C2N=C1OC)C=O)Br
Physical Data
AppearanceOff-white to light yellow powder
Melting Point, °C Solvent (Melting Point) 179 - 182
Spectra
No data available
Route of Synthesis (ROS)
Route of Synthesis (ROS) of 5-Quinoxalinecarboxaldehyde, 7-bromo-2,3-dimethoxy- CAS 187479-66-9
ConditionsYieldWith sodium In methanol; waterExperimental Procedure68.b bb 7-Bromo-5-formyl-2,3-dimethoxy-quinoxaline 1.38 g (60 mmol) of sodium are dissolved in portions, at 0° C. under N2, in 200 ml of methanol. At 0° C., 5.85 ml (65 mmol) of 2-nitropropane are added dropwise. 18.1 g (50 mmol) of 5-(bromomethyl)-7-bromo-2,3-dimethoxy-quinoxaline are then added. The beige suspension is heated to reflux and stirred for 1 hour. The reaction mixture is poured onto 600 ml of water and the methanol is distilled off. The residue is extracted twice with ethyl acetate, and the organic phases are dried over sodium sulfate and filtered with suction. The filtrate is concentrated and the residue is dried under a high vacuum. 7-Bromo-5-formyl-2,3-dimethoxy-quinoxaline is obtained in the form of beige crystals having a melting point of 179-182° C.
Safety and Hazards
GHS Hazard StatementsNot Classified
Other Data
TransportationUnder the room temperature and away from lightHS CodeStorageStorge at 2~8℃ and keep away from lightShelf Life2 yearsMarket Price
DruglikenessLipinski rules componentMolecular Weight297.108logP2.474HBA3HBD0Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)61.31Rotatable Bond (RotB)3Matching Veber Rules2
Use Pattern5-Quinoxalinecarboxaldehyde, 7-bromo-2,3-dimethoxy- CAS#: 187479-66-9 is mainly used as a pharmaceutical intermediate and building block for heterocyclic compound synthesis. https://www.chemwhat.com/5-quinoxalinecarboxaldehyde-7-bromo-23-dimethoxy-cas-187479-66-9/
Comments
Post a Comment