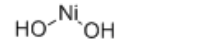
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
Product NameNickel HydroxideIUPAC Namenickel;dihydrate Molecular StructureCAS Registry Number 12054-48-7EINECS Number235-008-5MDL NumberMFCD00011140Beilstein Registry NumberSynonyms12054-48-7AKOS015903693CS-0089243dihydroxynickelEC 235-008-5EINECS 235-008-5HSDB 1827LS-96321MFCD00011140Nickel dihydroxideNickel hydroxide (II)Nickel hydroxide (Ni(OH)2)Nickel hydroxide (ous)Nickel Hydroxide nanowireNickel(2+) hydroxidenickel(II) dihydroxideNickel(II) hydroxide, for analysisnickel;dihydrateNickelous hydroxideUNII-L8UW92NW6JMolecular FormulaH4NiO2Molecular Weight94.724 InChIInChI=1S/Ni.2H2O/h;2*1H2 InChI KeyAIBQNUOBCRIENU-UHFFFAOYSA-N Canonical SMILESO.O.
Physical Data
AppearanceGreen powder
Spectra
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Temperature (IR Spectroscopy), °CBandssolid matrix-258
Description (UV/VIS Spectroscopy)Solvent (UV/VIS Spectroscopy)Comment (UV/VIS Spectroscopy)SpectrumsolidO4200 nm - 1730 nm
Route of Synthesis (ROS)
Route of Synthesis (ROS) of Nickel Hydroxide CAS 12054-48-7
ConditionsYieldIn ethanol; water at 150℃; for 2h; Microwave irradiation;95%In ethanol; water at 150℃; for 72h; Autoclave;89%
Safety and Hazards
Pictogram(s)SignalDangerGHS Hazard StatementsH302: Harmful if swallowed H315: Causes skin irritation H317: May cause an allergic skin reaction H332: Harmful if inhaled H334: May cause allergy or asthma symptoms or breathing difficulties if inhaled H341: Suspected of causing genetic defects H350i: May cause cancer by inhalation H360D ***: May damage the unborn child H372 **: Causes damage to organs through prolonged or repeated exposure H400: Very toxic to aquatic life H410: Very toxic to aquatic life with long lasting effects Precautionary Statement CodesP203, P260, P261, P264, P270, P271, P272, P273, P280, P281, P284, P301+P317, P302+P352, P304+P340, P317, P318, P319, P321, P330, P332+P317, P333+P313, P342+P316, P362+P364, P391, P405, and P501(The corresponding statement to each P-code can be found at the GHS Classification page.)
Other Data
TransportationStore at room temperature, sealed and away from light.HS CodeStorageStore at room temperature, sealed and away from light.Shelf Life1 yearMarket Price
DruglikenessLipinski rules componentMolecular Weight92.7047logP-0.934HBA2HBD2Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)40.46Rotatable Bond (RotB)0Matching Veber Rules2
Toxicity/Safety PharmacologyQuantitative Results
Use PatternNickel Hydroxide CAS#: 12054-48-7 is widely used as a positive electrode material in nickel-metal hydride (NiMH) batteries and nickel-cadmium (NiCd) batteries. It exhibits high energy storage density and longer cycle life, making these batteries widely used in portable electronic devices, power tools, and hybrid electric vehicles.
https://www.chemwhat.com/nickel-hydroxide-cas-12054-48-7/
Comments
Post a Comment