
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
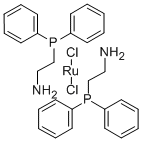
Product NameDichlorobis(2-(diphenylphosphino)ethylamine)ruthenium(II) IUPAC Namedichlororuthenium;2-diphenylphosphanylethanamineMolecular StructureCAS Registry Number 506417-41-0Synonyms506417-41-0Dichlorobis(2-(diphenylphosphino)ethylamine)ruthenium(II)Dichlorobisruthenium(II)dichlororuthenium;2-diphenylphosphanylethanamineSCHEMBL1767611DTXSID30584787MFCD07782004AKOS015900275DB-216527F728762-(Diphenylphosphanyl)ethanamine-dichlororuthenium (2:1)dichlorobisruthenium (ii)2-(Diphenylphosphanyl)ethan-1-amine--dichlororuthenium (2/1)Dichlorobis(2-(diphenylphosphino)ethylamine)ruthenium(II), 95%DICHLOROBISRUTHENIUMMolecular FormulaC28H32Cl2N2P2RuMolecular Weight630.5InChIInChI=1S/2C14H16NP.2ClH.Ru/c2*15-11-12-16(13-7-3-1-4-8-13)14-9-5-2-6-10-14;;;/h2*1-10H,11-12,15H2;2*1H;/q;;;;+2/p-2InChI KeyBJNVYFXBTNBKRU-UHFFFAOYSA-LSMILESC1=CC=C(C=C1)P(CCN)C2=CC=CC=C2.C1=CC=C(C=C1)P(CCN)C2=CC=CC=C2.ClCl
Patent InformationPatent IDTitlePublication DateUS2018/312454PROCESS FOR THE PREPARATION OF DEUTERATED ETHANOL FROM D22018
Physical Data
AppearanceYellow powder
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzChemical shifts31Pdichloromethane-d227161.839Chemical shifts31Pdichloromethane-d227161.8Chemical shifts, Spectrum31P1,4-dioxane-d830162
Route of Synthesis (ROS)
Route of Synthesis (ROS) of Dichlorobis(2-(diphenylphosphino)ethylamine)ruthenium(II) CAS 506417-41-0
ConditionsYieldIn toluene at 100℃; for 6h; Glovebox; Schlenk technique;Experimental Procedure2 0Preparation of (2)In a glove box, (0.25 mmol) was added to a schlenk flask equipped with a magnetic stir bar. The flask was then attached to a schlenk line and 3.4 mL of freshly distilled toluene added. The mixture was then rapidly stirred. A 1.7 mL toluene solution of Ph2PCH2CH2NH2, (0.50 mmol) in an NMR tube was then added via a cannula. Any residue in the NMR tube and cannula was washed into the flask with 1.7 mL of toluene. The light yellow mixture was then heated at 100° C. for 6 h. The yellow suspension that resulted was allowed to cool to RT before collecting the precipitate by filtration under Argon. The precipitate was then washed with 10.0 mL portions of toluene, three times (until colorless). The yellow solid was then dried in vacuo. Yield: 90%. Note: Excessive scraping of the product should be minimised to prevent the build-up of static electricity90%In toluene at 100℃; for 6h; Schlenk technique;90%
Safety and Hazards
GHS Hazard StatementsNot Classified
Other Data
DruglikenessLipinski rules componentMolecular Weight630.499HBA2HBD0Matching Lipinski Rules2Veber rules componentPolar Surface Area (PSA)0Rotatable Bond (RotB)4Matching Veber Rules2
Use PatternDichlorobis(2-(diphenylphosphino)ethylamine)ruthenium(II) CAS#: 506417-41-0 is primarily used as a homogeneous catalyst in hydrogenation, isomerization, and carbon–carbon bond-forming reactions. https://www.chemwhat.com/dichlorobis2-diphenylphosphinoethyla-cas-506417-41-0/
Comments
Post a Comment