
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
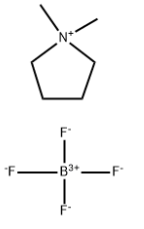
Product NamePyrrolidinium, 1,1-dimethyl-, tetrafluoroborateIUPAC Name1,1-dimethylpyrrolidin-1-ium;tetrafluoroborateMolecular StructureCAS Registry Number 69444-51-5Synonyms69444-51-51,1-Dimethylpyrrolidinium tetrafluoroborate834-341-91,1-Dimethylpyrrolidin-1-ium tetrafluoroborateSCHEMBL1448477n,n-dimethylpyrrolidinium tetrafluoroborateMolecular FormulaC6H14BF4NMolecular Weight186.99InChIInChI=1S/C6H14N.BF4/c1-7(2)5-3-4-6-7;2-1(3,4)5/h3-6H2,1-2H3;/q+1;-1InChI KeyQVXREJQVKRNKMK-UHFFFAOYSA-NSMILES(F)(F)(F)F.C1(CCCC1)C
Patent InformationPatent IDTitlePublication DateKR102774514METHOD FOR PREPARING HIGH PURITY QUATERNARY AMMONIUM SALT2025KR2015/79403One-pot water-free ionic liquids synthesis using trialkyl orthoesters2015
Physical Data
AppearanceColorless or light yellow transparent liquid
Melting Point, °C 340
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Frequency (NMR Spectroscopy), MHzChemical shifts19Fwater-d2Chemical shifts, Spectrum1Hwater-d2400Chemical shifts1Hdimethylsulfoxide-d6Chemical shifts1Hwater-d2500Chemical shifts19Fwater-d2Chemical shifts1Hwater-d2Chemical shifts13Cwater-d2
Route of Synthesis (ROS)
Route of Synthesis (ROS) of Pyrrolidinium, 1,1-dimethyl-, tetrafluoroborate CAS 69444-51-5
ConditionsYieldWith potassium hydroxide In acetonitrile at 55℃; for 13h; Cooling with ice;Experimental Procedure2 Example 2Put potassium hydroxide (7.954g) and sodium tetrafluoroborate (19.069g) into a two-necked round bottom flask containing 120ml of anhydrous acetonitrile, add 1,4-dichlorobutane (12.56g) under stirring, and finally Add dimethylamine (5.23g) dropwise to the above solution under ice bath conditions (the dripping time is 0.5h). After the dripping is completed, the reaction solution is stirred for 1h to uniformity, and the heating is turned on and the temperature is raised to 55°C for 12h to react. After completion, the reaction solution was returned to room temperature (20°C). The solvent in the reaction solution was removed from the above reaction solution by a rotary evaporator to obtain a powder-solid mixture. 50ml of absolute ethanol was added to the powder-solid mixture and stirred at 50°C for 1 hour. After cooling to room temperature, the impurity salt was removed by filtration to obtain an ethanol solution of N,N-dimethylpyrrolidinium tetrafluoroborate. The ethanol solution of N,N-dimethylpyrrolidinium tetrafluoroborate Recrystallize at low temperature (-20), then filter to obtain high-purity N,N-dimethylpyrrolidinium tetrafluoroborate, repeat recrystallization three times to obtain pure N,N-dimethyl Pyrrolidinium tetrafluoroborate, ethanol used for recrystallization is recovered for recrystallization of the next batch of materials. The obtained pure N,N-dimethylpyrrolidinium tetrafluoroborate was dried under vacuum at 110 for 12h to obtain dry pure N,N-dimethylpyrrolidinium tetrafluoroborate. Statistics , The yield is 91%.91%
Safety and Hazards
Pictogram(s)SignalDangerGHS Hazard StatementsH301 (50%): Toxic if swallowed H319 (50%): Causes serious eye irritation H412 (50%): Harmful to aquatic life with long lasting effects Precautionary Statement CodesP264, P264+P265, P270, P273, P280, P301+P316, P305+P351+P338, P321, P330, P337+P317, P405, and P501
Other Data
TransportationUnder the room temperature and away from lightHS CodeStorageUnder the room temperature and away from lightShelf Life1 yearMarket Price
DruglikenessLipinski rules componentMolecular Weight186.988HBA0HBD0Matching Lipinski Rules3Veber rules componentPolar Surface Area (PSA)0Rotatable Bond (RotB)0Matching Veber Rules2
Use PatternPyrrolidinium, 1,1-dimethyl-, tetrafluoroborate CAS#: 69444-51-5 is primarily used as an ionic liquid or electrolyte component in electrochemical applications such as batteries, capacitors, and electroplating. https://www.chemwhat.com/pyrrolidinium-11-dimethyl-tetrafluoroborate-cas-69444-51-5/
Comments
Post a Comment