
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
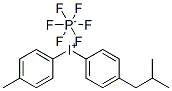
Product Name(4-Methylphenyl) iodonium hexafluorophosphateIUPAC Name(4-methylphenyl)-iodanium;hexafluorophosphateMolecular StructureCAS Registry Number 344562-80-7Synonyms344562-80-7Iodonium, (4-methylphenyl)-, hexafluorophosphate(1-)DTXSID7074905Iodonium, (4-methylphenyl)(4-(2-methylpropyl)phenyl)-, hexafluorophosphate(1-)Iodonium, (4-methylphenyl)(4-(2-methylpropyl)phenyl)-, hexafluorophosphate(1-) (1:1)Iodonium, (4-methylphenyl)-, hexafluorophosphate(1-) (1:1)DTXCID8038859(4-Methylphenyl) iodonium hexafluorophosphate(4-Isobutylphenyl)(p-tolyl)iodonium hexafluorophosphate(V)(4-methylphenyl)-iodanium;hexafluorophosphateSCHEMBL29351331YNDYCGZWQZEBCS-UHFFFAOYSA-NMFCD21604561DB-327830NS00078576G70508(4-methylphenyl)iodonium hexafluorophosphate(4-Isobutylphenyl)(p-tolyl)iodonium Hexafluorophosphate (ca. 70% in Propylene Carbonate)(4-methylphenyl)iodanium; hexafluoro--phosphanuide(4-methylphenyl)iodanium; hexafluoro-lambda-phosphanuide(4-Isobutylphenyl)(4-methylphenyl)iodonium Hexafluorophosphate (ca. 70% in Propylene Carbonate)Molecular FormulaC17H20F6IPMolecular Weight496.21InChIInChI=1S/C17H20I.F6P/c1-13(2)12-15-6-10-17(11-7-15)18-16-8-4-14(3)5-9-16;1-7(2,3,4,5)6/h4-11,13H,12H2,1-3H3;/q+1;-1InChI KeyYNDYCGZWQZEBCS-UHFFFAOYSA-NSMILESCC1=CC=C(C=C1)C2=CC=C(C=C2)CC(C)C.F(F)(F)(F)(F)F
Patent InformationPatent IDTitlePublication DateWO2023/195774DIPHENYLIODONIUM SALT FOR PHOTOINITIATOR AND PREPARATION METHOD THEREFOR2023
Physical Data
AppearanceColorless to yellowish liquid
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Chemical shifts1Hchloroform-d1
Description (IR Spectroscopy)Bands, Spectrum
Route of Synthesis (ROS)
Route of Synthesis (ROS) of (4-Methylphenyl) iodonium hexafluorophosphate CAS 344562-80-7
ConditionsYieldStage #1: 1-phenyl-2-methylpropane; 4-Iodotoluene With sodium persulfate; toluene-4-sulfonic acid at 20℃; for 20h;Stage #2: With potassium hexafluorophosphate for 10h;Experimental Procedure1-4 Example 4(1) 23.7 g of p-toluenesulfonic acid, 11.5 g of sodium persulfate, 10 g of p-iodotoluene and 7.2 g of isobutylbenzene were added to a reaction kettle in sequence and reacted at room temperature for 20 hours;(2) After adding 14 g of potassium hexafluorophosphate to the reaction solution obtained in step (1), the reaction was continued for 10 hours, washed twice with water and petroleum ether respectively, and concentrated to obtain a yellow viscous liquid with a yield of 87% and a purity of 99.27%.87%
Safety and Hazards
Pictogram(s)SignalDangerGHS Hazard StatementsH302 (100%): Harmful if swallowed H317 (100%): May cause an allergic skin reaction H318 (100%): Causes serious eye damage H373 (100%): May causes damage to organs through prolonged or repeated exposure H400 (100%): Very toxic to aquatic life H410 (100%): Very toxic to aquatic life with long lasting effects Precautionary Statement CodesP260, P261, P264, P264+P265, P270, P272, P273, P280, P301+P317, P302+P352, P305+P354+P338, P317, P319, P321, P330, P333+P317, P362+P364, P391, and P501
Other Data
DruglikenessLipinski rules componentMolecular Weight496.214HBA0HBD0Matching Lipinski Rules3Veber rules componentPolar Surface Area (PSA)0Rotatable Bond (RotB)4Matching Veber Rules2
Use Pattern4-Isobutylphenyl-4'-methylphenyliodonium hexafluorophosphate CAS 344562-80-7 is the most commonly used cationic photoinitiator, which can be sensitized by ITX and DETX to achieve longer absorption (LED curing) https://www.chemwhat.com/4-methylphenyl-4-2-methylpropylphenyl-iodonium-hexafluorophosphate-cas-344562-80-7/
Comments
Post a Comment