
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
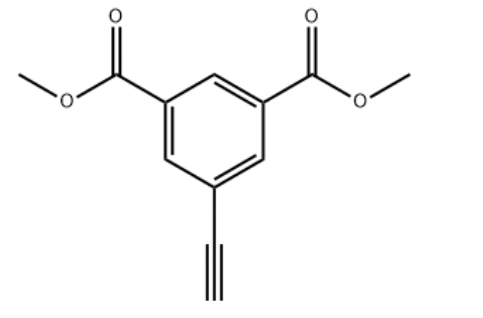
Product NameDimethyl 5-ethynylisophthalateIUPAC Namedimethyl 5-ethynylbenzene-1,3-dicarboxylateMolecular StructureCAS Registry Number 313648-56-5MDL NumberMFCD14635808SynonymsDimethyl 5-ethynylisophthalate313648-56-5dimethyl 5-ethynylbenzene-1,3-dicarboxylateMFCD14635808Dimethyl 5-ethynylbenzene-1,3-dioate1,3-DIMETHYL 5-ETHYNYLBENZENE-1,3-DICARBOXYLATEDimethyl5-ethynylisophthalateYSZC136SCHEMBL2065024AB9130Dimethyl 5-ethynylisophthalate, 97%BS-50456DB-319201CS-0110870EN300-11689002Molecular FormulaC12H10O4Molecular Weight218.2 InChIInChI=1S/C12H10O4/c1-4-8-5-9(11(13)15-2)7-10(6-8)12(14)16-3/h1,5-7H,2-3H3InChI KeyRSSODPUFUQKWIK-UHFFFAOYSA-NSMILESCOC(=O)C1=CC(=CC(=C1)C#C)C(=O)OC
Patent InformationPatent IDTitlePublication DateWO2019/117733THE USE OF ACETYLENE DERIVATIVES IN RUMINANTS2019
Physical Data
Melting Point, °C Solvent (Melting Point) 127 - 128134 - 136ethanol127 - 128124 - 125130 - 131
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzChemical shifts, Spectrum1Hchloroform-d124.84400Chemical shifts, Spectrum13Cchloroform-d124.84101DEPT (Distorsionless Enhancement by Polarisation Transfer), Chemical shifts, Spectrum13Cchloroform-d124.84100Chemical shifts, Spectrum13Cchloroform-d124.84100Chemical shifts, Spectrum1Hchloroform-d1500Chemical shifts13Cchloroform-d1125Chemical shifts1Hchloroform-d1500
Description (IR Spectroscopy)Solvent (IR Spectroscopy)BandspolyethyleneBandsnujol
Route of Synthesis (ROS)
Route-of-DiMethyl-5-ethynylisophthalate-CAS-313648-56-5
ConditionsYieldWith lithium aluminium hydride In tetrahydrofuran at 20℃;Experimental Procedure1.5-1 Step 5-1: Synthesis of e1:In a 250mL flask, weigh e0 (5.00g) and dissolve it in 100mL of THF, add LiAlH4 (2.0eq) under an ice bath, and then warm to room temperature to react overnight;The progress of the reaction is monitored by the dot plate, and the reaction is completely post-processed;The reaction solution was poured into 300 mL of ice water, stirred, suction filtered, washed with water, dried, and dried to obtain a bright yellow solid (3.2 g, 87.7% yield). The hydrogen spectrum of the compound is shown in Figure 8.87.7%
Safety and Hazards
GHS Hazard StatementsNot Classified
Other Data
TransportationUnder the room temperature and away from lightHS CodeStorageUnder the room temperature and away from lightShelf Life1 yearMarket Price
DruglikenessLipinski rules componentMolecular Weight218.209logP2.392HBA4HBD0Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)52.6Rotatable Bond (RotB)4Matching Veber Rules2
Use PatternLigand used in a synthesis of heterocycles by palladium-catalyzed C-N cross coupling of 3-bromothiophenes with 2-aminopyridines. Also used in a ruthenium-catalyzed alkylation of active methylene compounds with alcohols. Metal chelating ligand for catalysis. https://www.chemwhat.com/dimethyl-5-ethynylisophthalate-cas-313648-56-5/
Comments
Post a Comment