
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
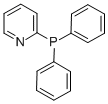
Product NameDiphenyl-2-pyridylphosphineIUPAC Namediphenyl(pyridin-2-yl)phosphaneMolecular StructureCAS Registry Number 37943-90-1MDL NumberMFCD00192108SynonymsDiphenyl-2-pyridylphosphine37943-90-12-(Diphenylphosphino)pyridineDTXSID00347462DTXCID30298534629-049-1diphenyl(pyridin-2-yl)phosphane2-Pyridyldiphenylphosphinediphenyl(2-pyridyl)phosphane2-Diphenylphosphanyl-pyridinediphenyl(2-pyridyl)phosphine2-(diphenylphosphanyl)pyridineMFCD001921084K685YSU7YDiphenyl 2-pyridyl phosphinediphenyl(2-pyridinyl)phosphineC17H14NP2-(Diphenylphosphino)pyridine; 2-Pyridyldiphenylphosphine; Diphenyl(2-pyridinyl)phosphine;diphenyl-2-pyridylphosphin2-pyridyl diphenylphosphineMolecular FormulaC17H14NPMolecular Weight263.27InChIInChI=1S/C17H14NP/c1-3-9-15(10-4-1)19(16-11-5-2-6-12-16)17-13-7-8-14-18-17/h1-14HInChI KeySVABQOITNJTVNJ-UHFFFAOYSA-N SMILESC1=CC=C(C=C1)P(C2=CC=CC=C2)C3=CC=CC=N3
Patent InformationPatent IDTitlePublication DateUS2024/79595NEW BENZAMIDE DERIVATIVES AS PPAR-GAMMA MODULATORS2024CN113698431CHROMENOPYRIDINE DERIVATIVES AS PHOSPHATIDYLINOSITOL PHOSPHATE KINASE INHIBITORS2021KR101920902A PET contrast compound for early diagnosis of cardiovascular diseases and use thereof2018US2013/274483ASYMMETRIC SYNTHESES FOR SPIRO-OXINDOLE COMPOUNDS USEFUL AS THERAPEUTIC AGENTS2013US2012/184738COPPER COMPLEXES FOR OPTOELECTRONIC APPLICATIONS2012WO2011/111806OD FOR PRODUCING α, β-UNSATURATED CARBOXYLATE, AND CATALYST FOR PRODUCING THEREOF2011US5326875Alkylation of azaglycine derivatives1994
Physical Data
AppearanceWhite to Off-white crystalline powder
Melting Point, °C Solvent (Melting Point) 84.2 - 85.584.2 - 85.5dichloromethane8582 - 838584 - 85aq. methanol
Boiling Point, °CPressure (Boiling Point), Torr1630.05
Description (Association (MCS))Solvent (Association (MCS))Partner (Association (MCS))NMR spectrum of the complexCD2Cl2BF4
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Frequency (NMR Spectroscopy), MHzChemical shifts, Spectrum1Hchloroform-d1chloroform-d131Pchloroform-d1Chemical shifts, Spectrum13Cchloroform-d1Chemical shifts, Spectrum31Pchloroform-d1Chemical shifts, Spectrum1Hchloroform-d1600Chemical shifts, Spectrum13Cchloroform-d1151Chemical shifts, Spectrum31Pchloroform-d1243
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Bands, SpectrumATR (attenuated total reflectance), SpectrumATR (attenuated total reflectance), Bandspotassium bromideBandsneat (no solvent, solid phase)Bands
Description (UV/VIS Spectroscopy)Solvent (UV/VIS Spectroscopy)Absorption Maxima (UV/VIS), nmExt./Abs. Coefficient, l·mol-1cm-1SpectrumdichloromethaneSpectrumSpectrumdichloromethane2589753Band assignment, Spectrumdichloromethane276Spectrumdichloromethane231, 261, 2938600, 6500, 1100
Route of Synthesis (ROS)
Route of Synthesis (ROS) of Diphenyl-2-pyridylphosphine CAS 37943-90-1
ConditionsYieldWith dihydrogen peroxide In dichloromethane; water at 20℃; for 3h; Glovebox; Inert atmosphere;98%With dihydrogen peroxide In dichloromethane; water at 20℃; for 3h; Glovebox; Inert atmosphere;Experimental ProcedureTris(4-methoxyphenyl)phosphine oxide (2a)General procedure: 1a (70.5 mg, 0.20 mmol), 4-phenylthioxanthone (3 mg, 0.01 mmol), CH3OH (30 mL) were added to a pyrex reaction flash which was equipped with a magnetic stirrer. The mixture was irradiated by a 23 W household lamp at rt under air atmosphere. The photoreaction was completed after 40 minutes as monitored by TLC (eluent: petroleum ether). The solvent was removed and the residue was purified by flash column chromatography on silica gel (eluent: petroleum ether/ethyl acetate = 10/1→EA) to afford 2a as a solid (74 mg, 100%); 1H NMR (400 MHz, CDCl3) δ 7.56 (dd, J = 11.6, 8.8 Hz, 6 H), 6.95 (dd, J = 8.8, 2.0 Hz, 6 H), 3.83 (s, 9 H).91%With hydrogen In ethyl acetate under 760.051 With dihydrogen peroxide In tetrahydrofuran; water for 0.533333h;88%
Safety and Hazards
Pictogram(s)SignalWarningGHS Hazard StatementsH315 (100%): Causes skin irritation H319 (100%): Causes serious eye irritation H335 (97.6%): May cause respiratory irritation H413 (95.1%): May cause long lasting harmful effects to aquatic life Precautionary Statement CodesP261, P264, P264+P265, P271, P273, P280, P302+P352, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P403+P233, P405, and P501(The corresponding statement to each P-code can be found at the GHS Classification page.)
Other Data
TransportationSlowly oxidized in the air for a long time, vacuum pack, and store in cold storageHS CodeStorageSlowly oxidized in the air for a long time, vacuum pack, and store in cold storageShelf Life1 year
DruglikenessLipinski rules componentMolecular Weight263.279logP4.78HBA1HBD0Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)26.48Rotatable Bond (RotB)3Matching Veber Rules2
Use PatternLigand in metal-catalyzed reactions:Carbonylation: Facilitates the introduction of carbonyl groups into organic molecules.Hydration: Aids in the addition of water to unsaturated compounds, forming alcohols or other functionalized derivatives.Dehydrogenative coupling: Promotes coupling reactions by removing hydrogen, often forming C-C or C-N bonds.Carbostannylation: Catalyzes the introduction of stannyl (Sn) groups into organic compounds.Dimethylstannylation: Specific variant of stannylation, introducing dimethylstannyl groups.Silylation: Facilitates the introduction of silyl groups, improving stability and reactivity of substrates.Reagent in Mitsunobu reactions:Used in the conversion of alcohols to various functional groups, such as esters, ethers, or amines, under mild conditions. https://www.chemwhat.com/diphenyl-2-pyridylphosphine-cas-37943-90-1/
Comments
Post a Comment