
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
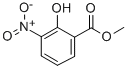
Product NameMETHYL 2-HYDROXY-3-NITROBENZOATEIUPAC Namepmethyl 2-hydroxy-3-nitrobenzoateMolecular StructureCAS Registry Number 22621-41-6EINECS Number923-922-3MDL NumberMFCD00272263SynonymsMethyl 2-hydroxy-3-nitrobenzoate22621-41-6Methyl 3-nitrosalicylate2-hydroxy-3-nitrobenzoic acid methyl esterMFCD00272263Benzoic acid, 2-hydroxy-3-nitro-, methyl ester3-Nitrosalicylic acid methyl esterDTXSID50344441Methyl 2-hydroxy-3-nitro-benzoateSalicylic acid, 3-nitro-, methyl ester2-(Methoxycarbonyl)-6-nitrophenol; 2-Hydroxy-3-nitrobenzoic Acid Methyl Ester; 3-Nitrosalicylic Acid Methyl Ester; Methyl 2-Hydroxy-3-nitrobenzoate; Methyl 3-NitrosalicylateMethyl-3-nitrosalicylatmethyl-m-nitrosalicylate2-hydroxy-3-nitro-benzoic acid methyl ester2-Hydroxy-3-nitro-benzoic acid, methyl esterSCHEMBL3912653-Nitro-salicylsauremethylesterDTXCID60295516Methyl 2-hydroxy-3-nitrobenzoate #Molecular FormulaC8H7NO5 Molecular Weight197.14InChIInChI=1S/C8H7NO5/c1-14-8(11)5-3-2-4-6(7(5)10)9(12)13/h2-4,10H,1H3InChI KeyNIBVYEHAFBEVFI-UHFFFAOYSA-NSMILESCOC(=O)C1=C(C(=CC=C1)(=O))O
Physical Data
AppearanceSticky yellow crystalline powder
Melting Point, °C Solvent (Melting Point) 129 - 13261 - 63130 - 131ethanol62142 - 143130 - 132128 - 130
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Frequency (NMR Spectroscopy), MHzChemical shifts1Hchloroform-d1Chemical shifts1Hdimethylsulfoxide-d6400Chemical shifts1Hchloroform-d1400Chemical shifts1Hchloroform-d1300Chemical shifts1HChemical shifts1Hchloroform-d1400Chemical shifts13Cchloroform-d1100
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Temperature (IR Spectroscopy), °CIRSpectrum
Route of Synthesis (ROS)
Route of Synthesis (ROS) of Methyl 3-nitrosalicylate CAS 22621-41-6
ConditionsYieldWith hydrogen; palladium on activated carbon In methanol at 20℃; under 3102.97 Torr; for 49h;Experimental Procedure68(2) Step 2 Methyl 3-Amino-2-hydroxybenzoateShake a suspension of methyl 2-hydroxy-3-nitrobenzoate (Step 1,18. 1 g, 93 mmol) and 5% palladium on carbon (5 g) in methanol (200 mL) at room temperature under hydrogen (60 psi) in a Parr bottle for 49 h. Filter the mixture through a plug of Celite and remove the filtrate solvent under reduced pressure to afford methyl 3-amino-2-hydroxybenzoate (Step 2) as a yellow solid (15.6 g, >99%) : 1H NMR (CDC13) 8 10.87 (s, 1H), 7.23 (dd, J = 7.7 Hz, 2H), 6.87 (t, J = 9.9 Hz, 1H), 3.95 (s, 3 H), 3.80 (br s, 2H) ; ESI MS m/z 168 +.99%With 10% Pd/C; hydrogen In tetrahydrofuran; ethanol; ethyl acetate at 20℃;Experimental Procedure51.51-1 Example 51-1; Synthesis of methyl 3-amino-2-hydroxybenzoateMethyl 2-hydroxy-3-nitrobenzoate (1.404 g, 7.12 mmol) in ethanol (70 ml), ethyl acetate (40 ml), was dissolved in THF (40 ml). It was stirred overnight at room temperature under a hydrogen atmosphere with 10% palladium-carbon thereto (140 mg). After the reaction, it was filtered through Celite and evaporated to give the title compound (1.174 g, 98.6%) as a brown solid.98.6%With hydrogen; palladium 10% on activated carbon In ethanol at 80℃; under 2625.26 Torr; for 2h;Experimental ProcedureTo a solution of 2-hydroxy-3-nitro-benzoic acid methyl ester (2.3 g, 11.67 mmol, 1.0 equiv) in ethanol (50 mL) was added palladium on activated charcoal 10% (0.47 g, 0.47 mmol, 0.04 equiv), the reaction vessel filled with hydrogen (3.5 bar) and stirred at 80° C. for 2 h. The catalyst was removed by filtration over celite and the solvent removed under reduced pressure yielding 1.9 g (95%) of the title compound which was used directly without further purification. 1H NMR (300 MHz, CDCl3): δ 3.88 (br s, 2H), 3.93 (s, 3H), 6.71 (t, J=7.9 Hz, 1H), 6.87 (dd, J=7.9 Hz, J=1.5 Hz, 1H), 7.24 (dd, J=7.9 Hz, J=1.5 Hz, 1H). 13C NMR (75 MHz, CDCl3): δ 52.13, 111.83, 118.71, 118.98, 119.55, 135.85, 149.70, 171.11.95%
Safety and Hazards
Pictogram(s)SignalWarningGHS Hazard StatementsH302 (20%): Harmful if swallowed H315 (100%): Causes skin irritation H319 (100%): Causes serious eye irritation H335 (100%): May cause respiratory irritation Precautionary Statement CodesP261, P264, P264+P265, P270, P271, P280, P301+P317, P302+P352, P304+P340, P305+P351+P338, P319, P321, P330, P332+P317, P337+P317, P362+P364, P403+P233, P405, and P501(The corresponding statement to each P-code can be found at the GHS Classification page.)
Other Data
TransportationUnder room temperature away from lightHS CodeStorageUnder room temperature away from lightShelf Life2 yearsMarket Price
Use PatternMETHYL 2-HYDROXY-3-NITROBENZOATE CAS#: 22621-41-6 can be used as an intermediate for pesticides, medicines, dyes, chemical fibers, rubber and energetic materials. https://www.chemwhat.com/methyl-2-hydroxy-3-nitrobenzoate-cas-22621-41-6/
Comments
Post a Comment