
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
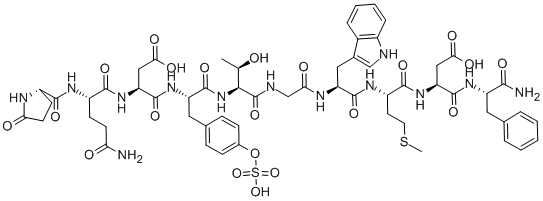
Product NameCAERULEINIUPAC Name(3S)-3-amino]pentanoyl]amino]-4-amino]-3-carboxy-1-oxopropan-2-yl]amino]-4-methylsulfanyl-1-oxobutan-2-yl]amino]-3-(1H-indol-3-yl)-1-oxopropan-2-yl]amino]-2-oxoethyl]amino]-3-hydroxy-1-oxobutan-2-yl]amino]-1-oxo-3-(4-sulfooxyphenyl)propan-2-yl]amino]-4-oxobutanoic acid Molecular StructureCAS Registry Number 17650-98-5MDL NumberMFCD00076478SynonymsCaeruleinCeruletide17650-98-5CeruleinCeruletidaCeruletidum5-Oxo-L-prolyl-L-glutaminyl-L-alpha-aspartyl-O-sulfo-L-tyrosyl-L-threonylglycyl-L-tryptophyl-L-methionyl-L-alpha-aspartyl-L-phenylalaninamide5-Oxo-L-prolyl-L-glutaminyl-L-aspartyl-L-tyrosyl-L-threonylglycyl-L-tryptophyl-L-methionyl-L-aspartylphenyl-L-alaninamide 4-(hydrogen sulfate) (ester)CHEBI:59219Caerulein (~85%)888Y08971BCeruletidum Ceruletida Ceruletide (3S)-3-{carbamoyl}-3-formamido}butanamido]-3-carboxypropanamido]-3-propanamido]-3-hydroxybutanamido]acetamido}-3-(1H-indol-3-yl)propanamido]-4-(methylsulfanyl)butanamido]propanoic acid5-oxo-L-prolyl-L-glutaminyl-L-a-aspartyl-O-sulfo-L-tyrosyl-L-threonylglycyl-L-tryptophyl-L-methionyl-L-a-aspartyl-L-phenylalaninamideCCRIS 3063Ceruletide (USAN/INN)UNII-888Y08971BCaerulein, sulfatedMFCD00076478CERULETIDE CERULETIDE CERULETIDE CERULETIDE CERULETIDE SCHEMBL29520GTPL7589CHEMBL1201355DTXSID8040434YRALAIOMGQZKOW-HYAOXDFASA-NHY-A0190AKOS030529560CS-5876DB00403AS-56061NS00004791C73362D03442EN300-23248418Q5065299pGlu-Gln-Asp-Tyr(SO3H)-Thr-Gly-Trp-Met-Asp-Phe-NH2(3S)-3-{carbamoyl}-3-formamido}butanamido]-3-carboxypropanamido]-3-propanamido]-3-hydroxybutanamido]acetamido}-3-(indol-3-yl)propanamido]-4-(methylsulfanyl)butanamido]propanoic acid5-OXO-L-PROLYL-L-GLUTAMINYL-L-A-ASPARTYL-O-SULFO-L-TYROSYL-L-THREONYLGLYCYL-L-TRYPTOPHYL-L-METHIONYL-L-.ALPHA.-ASPARTYL-L-PHENYLALANINAMIDEMolecular FormulaC58H73N13O21S2Molecular Weight1352.4InChIInChI=1S/C58H73N13O21S2/c1-29(72)49(71-57(87)40(23-31-12-14-33(15-13-31)92-94(89,90)91)68-56(86)43(26-48(78)79)69-52(82)37(16-18-44(59)73)65-51(81)36-17-19-45(74)63-36)58(88)62-28-46(75)64-41(24-32-27-61-35-11-7-6-10-34(32)35)54(84)66-38(20-21-93-2)53(83)70-42(25-47(76)77)55(85)67-39(50(60)80)22-30-8-4-3-5-9-30/h3-15,27,29,36-43,49,61,72H,16-26,28H2,1-2H3,(H2,59,73)(H2,60,80)(H,62,88)(H,63,74)(H,64,75)(H,65,81)(H,66,84)(H,67,85)(H,68,86)(H,69,82)(H,70,83)(H,71,87)(H,76,77)(H,78,79)(H,89,90,91)/t29-,36+,37+,38+,39+,40+,41+,42+,43+,49+/m1/s1InChI KeyYRALAIOMGQZKOW-HYAOXDFASA-NIsomeric SMILESC((C(=O)NCC(=O)N(CC1=CNC2=CC=CC=C21)C(=O)N(CCSC)C(=O)N(CC(=O)O)C(=O)N(CC3=CC=CC=C3)C(=O)N)NC(=O)(CC4=CC=C(C=C4)OS(=O)(=O)O)NC(=O)(CC(=O)O)NC(=O)(CCC(=O)N)NC(=O)5CCC(=O)N5)O
Patent InformationPatent IDTitlePublication DateUS4351829Use of polypeptides as analgesic drugs1982
Physical Data
AppearanceWhite Powder
Spectra
No data available
Route of Synthesis (ROS)
Route of Synthesis (ROS) of CAERULEIN CAS 17650-98-5
ConditionsYieldExperimental ProcedureXIII N-terminal tetrapeptide sulfate ester hydrazide of CRLbutyl ester and coupled with DCC to the Tyr-sulfate ester-resin. After deblocking with 50% TFA/CH2 Cl2 (v/v), BOC-Glu(p-nitrophenyl ester) and pGlu(pentafluorophenyl ester) are respectively used for stepwise coupling. The tetrapeptide sulfate ester is cleaved from the resin in DMF with 30-fold excess hydrazine and isolated by precipitation with ethyl ether. It is further purified by precipitation from DMF with ethylacetate, yielding a pure Caerulein 1-4 tetrapeptide hydrazide suitable for further synthesis.
Safety and Hazards
GHS Hazard StatementsNot Classified
Source: European Chemicals Agency (ECHA)License Note: Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: “Source: European Chemicals Agency, http://echa.europa.eu/”. Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page.License URL: https://echa.europa.eu/web/guest/legal-noticeRecord Name: (1-Cyano-2-ethoxy-2-oxoethylidenaminooxy)dimethylamino-morpholino-carbenium hexafluorophosphateURL: https://echa.europa.eu/information-on-chemicals/cl-inventory-database/-/discli/details/213446Description: The information provided here is aggregated from the “Notified classification and labelling” from ECHA’s C&L Inventory. Read more: https://echa.europa.eu/information-on-chemicals/cl-inventory-database
Other Data
TransportationStore at -20°C for long time, in container tightly sealed; Protect from light.HS CodeStorageStore at -20°C for long time, in container tightly sealed; Protect from light.Shelf Life2 yearsMarket Price
DruglikenessLipinski rules componentMolecular Weight1352.42logP-5.328HBA33HBD17Matching Lipinski Rules1Veber rules componentPolar Surface Area (PSA)585.08Rotatable Bond (RotB)47Matching Veber Rules0
Use PatternFrogletin (also known as Frog Dermaseptin) is a type of antimicrobial peptide extracted from frog skin, with a variety of biological activities. Its main functions include:Antimicrobial Activity: Frogletin has significant inhibitory or killing effects on various pathogens, including bacteria, fungi, and viruses. This gives it potential applications in fighting infections, especially those caused by multidrug-resistant bacteria.Antitumor Activity: Some studies have shown that Frogletin peptides can selectively kill certain cancer cells without significantly harming normal cells, making it a promising candidate for cancer treatment.Immune Modulation: Frogletin has the potential to regulate immune responses by stimulating or suppressing immune cell activity, enhancing the body’s defense against infections and diseases.Anti-inflammatory Effects: In certain cases, Frogletin exhibits anti-inflammatory properties, helping to reduce inflammation caused by infections or immune reactions. https://www.chemwhat.com/caerulein-cas-17650-98-5/
Comments
Post a Comment