IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
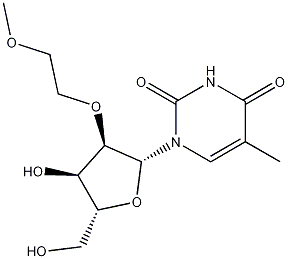
Product Name2'-O-(2-Methoxyethyl)-5-methyluridineIUPAC Name1--5-methylpyrimidine-2,4-dioneMolecular StructureCAS Registry Number 163759-49-7MDL NumberMFCD02682964Synonyms163759-49-72'-O-(2-Methoxyethyl)-5-methyl-uridine2'-O-(2-methoxyethyl)-5-methylUridineUridine, 2'-O-(2-methoxyethyl)-5-methyl-2'-O-MOE-5-Me-rU9Q36I4D9VB1--5-methylpyrimidine-2,4-dione1-((2R,3R,4R,5R)-4-hydroxy-5-(hydroxymethyl)-3-(2-methoxyethoxy)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione2'-MOE-5-MethyluridineUNII-9Q36I4D9VBSCHEMBL50826DTXSID104599642'O-Methoxyethyl-5-methyluridineNEVQCHBUJFYGQO-DNRKLUKYSA-N2'-o-methoxyethyl-5-methyluridine2'-O-Methoxvethyl-5-methyluridine2'--O-Methoxyethyl-5-methyluridineMFCD026829642'-O-(methoxyethyl)-5-methyluridine2'--O--Methoxyethyl-5-methyluridineAKOS016005091AM10114HY-W048488AC-24669BP-58846CS-16579CS-0100774F12191A851922J-7003091-((2R,3R,4R,5R)-3-(2-methoxyethoxy)-tetrahydro-4-hydroxy-5-(hydroxymethyl)furan-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dioneMolecular FormulaC13H20N2O7Molecular Weight316.31InChIInChI=1S/C13H20N2O7/c1-7-5-15(13(19)14-11(7)18)12-10(21-4-3-20-2)9(17)8(6-16)22-12/h5,8-10,12,16-17H,3-4,6H2,1-2H3,(H,14,18,19)/t8-,9-,10-,12-/m1/s1InChI KeyNEVQCHBUJFYGQO-DNRKLUKYSA-NIsomeric SMILESCC1=CN(C(=O)NC1=O)2(((O2)CO)O)OCCOC
Physical Data
AppearanceWhite powder
Melting Point, °C Solvent (Melting Point) 115.5 - 116.5ethyl acetate115.5 - 116.5115.5 - 116.5methanol, ethyl acetate
Spectra
Route of Synthesis (ROS)
Route of Synthesis (ROS) of 2'-O-(2-Methoxyethyl)-5-methyluridine CAS 163759-49-7
ConditionsYieldStage #1: O-2,2'-cyclo-5-methyluridine; tris(2-methoxyethyl)borate With sodium hydrogencarbonate In 2-methoxy-ethanol at 130℃; for 21h;Stage #2: With water for 0.5h; Heating / reflux;Experimental Procedure2,2'-Anhydro-5-methyl-uridine (2000 g, 8.32 mol), tris(2-methoxyethyl)borate (2504 g, 10.60 mol), sodium bicarbonate (60 g, 0.70 mol) and anhydrous 2-methoxyethanol (5 L) were combined in a 12 L three necked flask and heated to 130° C. (internal temp) at atmospheric pressure, under an argon atmosphere with stirring for 21 h. TLC indicated a complete reaction. The solvent was removed under reduced pressure until a sticky gum formed (50-85° C. bath temp and 100-11 mm Hg) and the residue was redissolved in water (3 L) and heated to boiling for 30 min in order the hydrolyze the borate esters. The water was removed under reduced pressure until a foam began to form and then the process was repeated. HPLC indicated about 77% product, 15% dimer (5' of product attached to 2' of starting material) and unknown derivatives, and the balance was a single unresolved early eluting peak. The gum was redissolved in brine (3 L), and the flask was rinsed with additional brine (3 L). The combined aqueous solutions were extracted with chloroform (20 L) in a heavier-than continuous extractor for 70 h. The chloroform layer was concentrated by rotary evaporation in a 20 L flask to a sticky foam (2400 g). This was coevaporated with MeOH (400 mL) and EtOAc (8 L) at 75° C. and 0.65 atm until the foam dissolved at which point the vacuum was lowered to about 0.5 atm. After 2.5 L of distillate was collected a precipitate began to form and the flask was removed from the rotary evaporator and stirred until the suspension reached ambient temperature. 69.4%
Safety and Hazards
No data available
Other Data
TransportationStorage at -20°,keep away from light and sealedHS CodeStorageStorage at -20°,keep away from light and sealedShelf Life1 yearMarket Price
DruglikenessLipinski rules componentMolecular Weight316.311logP-2.036HBA9HBD3Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)117.56Rotatable Bond (RotB)6Matching Veber Rules2
Use Pattern3-Aminopyridine CAS#: 462-08-8 is an intermediate in pesticides and dyes; pesticide raw materials; analytical reagents.
https://www.chemwhat.com/2-o-2-methoxyethyl-5-methyluridine-cas-163759-49-7/
Comments
Post a Comment