IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
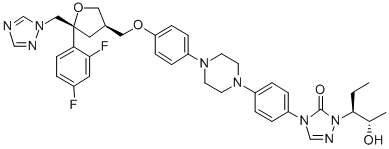
Product NamePosaconazole IUPAC Name4-methoxy]phenyl]piperazin-1-yl]phenyl]-2--1,2,4-triazol-3-one Molecular StructureCAS Registry Number 171228-49-2MDL NumberMFCD00941162SynonymsPosaconazole171228-49-2NoxafilSch 56592SCH-56592Posaconazole SPSCH56592Schering 56592(-)-POSACONAZOLEUNII-6TK1G07BHZ4-methoxy]phenyl]piperazin-1-yl]phenyl]-2--1,2,4-triazol-3-one6TK1G07BHZ4-(4-(4-(4-(((3R,5R)-5-((1H-1,2,4-TRIAZOL-1-YL)METHYL)-5-(2,4-DIFLUOROPHENYL)TETRAHYDROFURAN-3-YL)METHOXY)PHENYL)PIPERAZIN-1-YL)PHENYL)-1-((2S,3S)-2-HYDROXYPENTAN-3-YL)-1H-1,2,4-TRIAZOL-5(4H)-ONEDTXSID6049066CHEBI:64355HSDB 7421CHEMBL1397DTXCID401474254Molecular FormulaC37H42F2N8O4Molecular Weight700.8InChIInChI=1S/C37H42F2N8O4/c1-3-35(26(2)48)47-36(49)46(25-42-47)31-7-5-29(6-8-31)43-14-16-44(17-15-43)30-9-11-32(12-10-30)50-20-27-19-37(51-21-27,22-45-24-40-23-41-45)33-13-4-28(38)18-34(33)39/h4-13,18,23-27,35,48H,3,14-17,19-22H2,1-2H3/t26-,27+,35-,37-/m0/s1 InChI KeyRAGOYPUPXAKGKH-XAKZXMRKSA-NIsomeric SMILESCC((C)O)N1C(=O)N(C=N1)C2=CC=C(C=C2)N3CCN(CC3)C4=CC=C(C=C4)OC5C(OC5)(CN6C=NC=N6)C7=C(C=C(C=C7)F)F
Patent InformationPatent IDTitlePublication DateWO2019/40363AZOLE ANALOGUES AND METHODS OF USE THEREOF2019US2019/388397COMPOSITIONS AND METHODS FOR THE TREATMENT AND PREVENTION OF NEUROLOGICAL DISORDERS 2019US2014/303184PROCESS FOR THE PREPARATION OF A CHIRAL COMPOUND2014US2014/343285Process for the Preparation of Triazole Antifungal Drug, Its Intermediates and Polymorphs Thereof2014
Physical Data
Appearancewhite to off-white powder
Melting Point, °C Solvent (Melting Point) 168.2171.44168 - 171171.4acetone, water170.1171
Density, g·cm-3Measurement Temperature, °C1.321-173.16
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)MAS (Magic-Angle Spinning), Spectrum1H2D-NMR, Spectrum1H, 13CMAS (Magic-Angle Spinning), Spectrum13CChemical shifts, Spectrum1HChemical shifts, Spectrum1Hdimethylsulfoxide-d6
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Temperature (IR Spectroscopy), °CBandspotassium bromide Bands, Spectrum
Description (UV/VIS Spectroscopy)Solvent (UV/VIS Spectroscopy)Absorption Maxima (UV/VIS), nmUV excited state absorption, Spectrummethanol203
Route of Synthesis (ROS)
Route of Synthesis (ROS) of Posaconazole CAS 171228-49-2
ConditionsYieldWith hydrogenchloride; water at 25 - 63℃; Temperature;95%With hydrogenchloride; Pd/C; hydrogen In methanol; water at 50℃; under 750.075 Torr; for 2h; Temperature; Reagent/catalyst; Pressure; Sealed tube; Large scale;93.6%With hydrogenchloride In water at 45 - 50℃; for 20h; Large scale;Experimental ProcedureAdd 95.5kg of concentrated hydrochloric acid to a 300L reactor, The compound 3 obtained in the previous step was added with stirring, and then the temperature was raised to 45-50°C to react for 20 hours. TLC (EA) detection showed that there was no spot on compound 3. The reaction is considered complete. After the reaction is complete, cool down to 20-30°C, Use 20%-30% sodium hydroxide solution 100.0-120.0kg to adjust PH to 9-10, temperature control≤30, crystallize at room temperature for 2h after completion, filter, The filter cake is rinsed with 10kg methanol, and the filter cake is air-dried at 40-45°C for 8 hours . Get 12.12kg. The yield was 84.43%. H-HNM see Figure 3. Refining of posaconazole 11.4kg of crude posaconazole was added to 90kg of methanol, heated to reflux for 30min, and 67.5kg of purified water was added dropwise while keeping warm. Cool down naturally to 20-25, keep incubating and crystallize for 2h, filter, rinse with 10kg of filter cake and methanol/water mixed solution, add ratio of filter cake to methanol/water mixed solution at 1:1, 40-45 blast drying The finished product was 10.48kg in 8h. The yield was 91.93%.91.93%
Safety and Hazards
Pictogram(s)SignalDangerGHS Hazard StatementsH319 (75%): Causes serious eye irritation H351 (20.83%): Suspected of causing cancer H361 (97.92%): Suspected of damaging fertility or the unborn child H372 (100%): Causes damage to organs through prolonged or repeated exposure H400 (95.83%): Very toxic to aquatic life H410 (95.83%): Very toxic to aquatic life with long lasting effects Precautionary Statement CodesP203, P260, P264, P264+P265, P270, P273, P280, P305+P351+P338, P318, P319, P337+P317, P391, P405, and P501(The corresponding statement to each P-code can be found at the GHS Classification page.)
Other Data
TransportationUnder room temperature away from lightHS CodeStorageUnder room temperature away from lightShelf Life1 yearMarket Price
DruglikenessLipinski rules componentMolecular Weight700.788logP5.721HBA11HBD1Matching Lipinski Rules1Veber rules componentPolar Surface Area (PSA)111.79Rotatable Bond (RotB)12Matching Veber Rules1
Quantitative Results1 of 2,228Comment (Pharmacological Data)Bioactivities presentReferenceCrystalline antifungal polymorph2 of 2,228Comment (Pharmacological Data)Bioactivities presentReferenceActivity of SCH 56592 compared with those of fluconazole and itraconazole against Candida spp. 3 of 2,228Comment (Pharmacological Data)Bioactivities presentReferenceIn vitro activities of posaconazole, itraconazole, voriconazole, amphotericin B, and fluconazole against 37 clinical isolates of zygomycetes4 of 2,228Comment (Pharmacological Data)Bioactivities presentReferenceIn vivo activity of posaconazole against Mucor spp. in an immunosuppressed-mouse model.5 of 2,228Comment (Pharmacological Data)Bioactivities presentReferenceIn vitro activities of posaconazole (Sch 56592) compared with those of itraconazole and fluconazole against 3,685 clinical isolates of Candida spp. and Cryptococcus neoformans6 of 2,228Comment (Pharmacological Data)Bioactivities presentReferenceCRYSTALLINE ANTIFUNGAL POLYMORPH7 of 2,228Comment (Pharmacological Data)Bioactivities presentReferencePROCESS FOR PREPARING POSACONAZOLE AND INTERMEDIATES THEREOF8 of 2,228Comment (Pharmacological Data)Bioactivities presentReferenceNew antifungal peptides. Synthesis, bioassays and initial structure prediction by CD spectroscopy9 of 2,228Comment (Pharmacological Data)Bioactivities presentReferenceAnti-leishmanial activity of betulin derivatives10 of 2,228Comment (Pharmacological Data)Bioactivities presentReferenceSteroid-transforming enzymes in fungi
Use PatternPosaconazole CAS#: 171228-49-2 is a second-generation triazole antifungal drug widely used for the prevention and treatment of invasive fungal infections.
https://www.chemwhat.com/posaconazole-cas-171228-49-2/
Comments
Post a Comment