IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
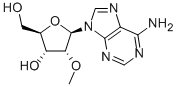
Product Name2'-O-MethyladenosineIUPAC Name(2R,3R,4R,5R)-5-(6-aminopurin-9-yl)-2-(hydroxymethyl)-4-methoxyoxolan-3-olMolecular StructureCAS Registry Number 2140-79-6MDL NumberMFCD00056002Synonyms2'-O-Methyladenosine2140-79-6Adenosine, 2'-O-methyl-(2R,3R,4R,5R)-5-(6-amino-9H-purin-9-yl)-2-(hydroxymethyl)-4-methoxytetrahydrofuran-3-ol2'-O-Methyl adenosine(2R,3R,4R,5R)-5-(6-aminopurin-9-yl)-2-(hydroxymethyl)-4-methoxyoxolan-3-olCHEMBL732372'-O-METHYL-ADENOSINE02YX82IHZ5CHEBI:69426cordysinin BMFCD00056002C11H15N5O4UNII-02YX82IHZ5SCHEMBL259362'-O-Me-ADTXSID50175671BCP17411BDBM50144945HG1274PDSP1_001054PDSP2_001038AKOS016003815AC-8216CS-W012268HY-W011552(2R,3R,4R,5R)-5-(6-aminopurin-9-yl)-2-(hydroxymethyl)-4-methoxy-tetrahydrofuran-3-olBP-58835DS-14586PD018166Q633928555-(6-amino-9H-purin-9-yl)-2-(hydroxymethyl)-4-methoxyoxolan-3-olMolecular FormulaC11H15N5O4Molecular Weight281.27InChIInChI=1S/C11H15N5O4/c1-19-8-7(18)5(2-17)20-11(8)16-4-15-6-9(12)13-3-14-10(6)16/h3-5,7-8,11,17-18H,2H2,1H3,(H2,12,13,14)/t5-,7-,8-,11-/m1/s1 InChI KeyFPUGCISOLXNPPC-IOSLPCCCSA-NIsomeric SMILESCO1((O1N2C=NC3=C(N=CN=C32)N)CO)O
Physical Data
AppearanceWhite powder
Melting Point, °C Solvent (Melting Point) 204 - 206202 - 203ethanol200 - 201198 - 201203 - 207
Density, g·cm-31.482
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzChemical shifts, Spectrum1H25Chemical shifts1Hdimethylsulfoxide-d625500Chemical shifts13Cdimethylsulfoxide-d6125Chemical shifts1HCD3OD500
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Comment (IR Spectroscopy)Bands neat (no solvent, solid phase)Bandspotassium bromide
Description (UV/VIS Spectroscopy)Solvent (UV/VIS Spectroscopy)Comment (UV/VIS Spectroscopy)Absorption Maxima (UV/VIS), nmExt./Abs. Coefficient, l·mol-1cm-1Absorption maximaH2O25914000Absorption maxima259400Absorption maximaH2ORemark: pH 725913804
Route of Synthesis (ROS)
Route of Synthesis (ROS) of 2'-O-Methyladenosine CAS 2140-79-6
ConditionsYieldWith 1H-imidazole In pyridine at 65℃; for 12h; silylation;93%With 1H-imidazole; dmap In N,N-dimethyl-formamide at 20℃; for 28h;83%With pyridine; 1H-imidazole at 20℃; for 15h;Experimental Procedure117-130 Preparation of compound 7-2:To a solution of 7-1 (15.0 g, 53.3 mmol) in dry pyridine (150 mL) was added TBSC1 (20.0 g, 133.3 mmol) and Imidazole (10.8 g, 159.9 mmol). The mixture was stirred at r.t. for l5h. TLC showed 7-1 was consumed completely. The reaction mixture was concentrated in vacuo to give residue. The residue was quenched with DCM (500 mL). The DCM layer was washed with H2O (1 L*2) 2 times and brine. The DCM layer concentrated in vacuo to give crude 7-2 (27.2 g, 53.3 mmol) as a yellow oil. The crude 7-2 was used in next step directly. ESI-LCMS m/z 510.5 +.99%
Safety and Hazards
No data available
Other Data
No data available
DruglikenessLipinski rules componentMolecular Weight281.271logP-1.327HBA9HBD3Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)128.54Rotatable Bond (RotB)3Matching Veber Rules2
Quantitative Results1 of 25Comment (Pharmacological Data)Bioactivities presentReferenceNovel adenosine derivatives and pharmaceutical composition containing them as an active ingredient2 of 25Comment (Pharmacological Data)Bioactivities presentReferenceMethylation of Adenosine and Related Nucleosides with Trimethylselenonium Hydroxyde, and Regiospecific Effects of Copper(II) Ions 3 of 25Comment (Pharmacological Data)Bioactivities presentReferenceNucleic acid related compounds. 36. Synthesis of the 2'-O-methyl and 3'-O-methyl ethers of guanosine and 2-aminoguanosine and correlation of O'-methylnucleoside 13C nmr spectral shifts4 of 25Comment (Pharmacological Data)Bioactivities presentReferenceThe first synthesis of herbicidin B. stereoselective construction of the tricyclic undecose moiety by a conformational restriction strategy using steric repulsion between adjacent bulky silyl protecting groups on a pyranose ring5 of 25Comment (Pharmacological Data)Bioactivities presentReferenceInhibitory actvity of the compound against Fatty acid amide hydrolase in 0.1 M sodium phosphate, pH 8.06 of 25Comment (Pharmacological Data)Bioactivities presentReferenceRecognition and detection of 8-oxo-rG in RNA using the DNA/OMeRNA chimera probes containing fluorescent adenosine-diazaphenoxazine analog7 of 25Comment (Pharmacological Data)Bioactivities presentReferenceNucleoside derivatives as inhibitors of RNA-dependent RNA viral polymerase8 of 25Comment (Pharmacological Data)Bioactivities presentReferenceRNAi agents and compositions for inhibiting expression of apolipoprotein C-III (APOC3)9 of 25Comment (Pharmacological Data)Bioactivities presentReferenceMODIFIED SHORT INTERFERING NUCLEIC ACID (SINA) MOLECULES AND USES THEREOF10 of 25Comment (Pharmacological Data)Bioactivities presentReferenceAdolescent alcohol exposure changes RNA modifications in adult brain by mass spectrometry-based comprehensive profiling analysis
Use Pattern2'-O-Methyladenosine CAS#: 2140-79-6 (N-EPSILON-L-LYSINE METHYL ESTER HYDROCHLORIDE SALT and it is used in IVD.
https://www.chemwhat.com/2-o-methyladenosine-cas-2140-79-6/
Comments
Post a Comment