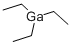
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
Product NameTRIETHYLGALLIUMIUPAC Nametriethylgallane Molecular StructureCAS Registry Number 1115-99-7EINECS Number214-232-7SynonymsTriethylgalliumtriethylgallane1115-99-7Gallium, triethyl-EINECS 214-232-7MFCD00015094Triethylgallium, elec. gr.DTXSID1061497AKOS015914832FT-0654071NS00081294A802388Q2346835Molecular FormulaC6H15GaMolecular Weight156.91InChIInChI=1S/3C2H5.Ga/c3*1-2;/h3*1H2,2H3InChI KeyRGGPNXQUMRMPRA-UHFFFAOYSA-NIsomeric SMILESCC(CC)CC
Patent InformationPatent IDTitlePublication Date30962119PROCESS FOR THE PREPARATION OF TRIMETHYL METAL COMPOUNDS201737457058High-purity trialkylgallium and its manufacturing method (by machine translation)201526873185METHODS OF PRODUCING TRIMETHYLGALLIUM201423677215PROCESS FOR PREPARING TRIALKYLGALLIUM COMPOUNDS2013
Physical Data
No data available
Melting Point, °C -82-82.3
Boiling Point, °CPressure (Boiling Point), Torr10976 - 786034 - 35103410130 - 13565014257 - 63256
Density, g·cm-3Measurement Temperature, °C1.317-140.161.307-821.331-1451.057630
Description (Association (MCS))Partner (Association (MCS))GaAs(100)AdsorptionGaAs(100)AdsorptionN2H4-covered MgO(100)AdsorptionGaAs#dotZnAdsorptionAl/GaAs
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzChemical shifts, Spectrum1Hbenzene-d6400.1Chemical shifts, Spectrum13Cbenzene-d6100.61Hbenzene-d61HCyclohexane-d1225
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Comment (IR Spectroscopy)Spectrumneat (no solvent, gas phase) 400 cm**-1 - 4000 cm**-1IR
Description (UV/VIS Spectroscopy)Solvent (UV/VIS Spectroscopy)Comment (UV/VIS Spectroscopy)Spectrumgaseous matrix190 nm - 390 nm
Route of Synthesis (ROS)
Route of Synthesis (ROS) of TRIETHYLGALLIUM CAS 1115-99-7
ConditionsYieldWith potassium chloride; sodium chloride at 120 - 130℃; under 225.023 Torr; Pressure; Concentration; Temperature; Inert atmosphere;A 75.9%B n/aWith potassium chloride; sodium chloride at 120℃; under 225.023 Torr; Inert atmosphere;Experimental Procedure4 Preparation of triethylgallium (TEG)Example 4 Preparation of triethylgallium (TEG) 68.9 g (0.39 mol) of GaCI3, 32.0 g (0.55 mol, 1 .4 equivalents) of dry NaCI and 17.5 g (0.23 mol, 0.6 equivalent) of dry KCI are placed under argon in a 500 ml four-neck flask provided with stirrer, dropping funnel and a separator maintained at 130°C. While stirring, 120.2 g (0.47 mol, 1 .2 equivalents) of ethylaluminium sesquichloride (Et3AI2Cl3) are added in such a way that the temperature in the reaction mixture does not exceed 120°C. The reaction mixture is subsequently heated while applying a reduced pressure of 300 mbar, and Et3Ga is thus isolated (46.6 g; direct yield 75.9%). A 75.9%B 19%
Safety and Hazards
Pictogram(s)SignalDangerGHS Hazard StatementsH225 (27.54%): Highly Flammable liquid and vapor H250 (100%): Catches fire spontaneously if exposed to air H260 (71.74%): In contact with water releases flammable gases which may ignite spontaneously H261 (27.54%): In contact with water releases flammable gas H314 (100%): Causes severe skin burns and eye damage H318 (81.16%): Causes serious eye damage Precautionary Statement CodesP210, P222, P223, P231, P231+P232, P233, P240, P241, P242, P243, P260, P264, P264+P265, P280, P301+P330+P331, P302+P335+P334, P302+P361+P354, P303+P361+P353, P304+P340, P305+P354+P338, P316, P317, P321, P363, P370+P378, P402+P404, P403+P235, P405, and P501(The corresponding statement to each P-code can be found at the GHS Classification page.)
Other Data
No data available
DruglikenessLipinski rules componentMolecular Weight156.908logP2.658HBA0HBD0Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)0Rotatable Bond (RotB)3Matching Veber Rules2
Use PatternTRIETHYLGALLIUM CAS#: 1115-99-7 is a commonly used precursor for the production of semiconductor materials such as gallium nitride (GaN) and gallium arsenide (GaAs). These semiconductor materials find critical applications in electronic devices, lasers, and optoelectronic devices. And semiconductor materials derived from triethylgallium can be utilized in the manufacturing of optoelectronic devices such as light-emitting diodes (LEDs), laser diodes (LDs), etc.
https://www.chemwhat.com/triethylgallium-cas-1115-99-7/
Comments
Post a Comment