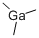
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
Product NameTRIMETHYLGALLIUMIUPAC Nametrimethylgallane Molecular StructureCAS Registry Number 1445-79-0EINECS Number215-897-6MDL NumberMFCD00014841SynonymsTRIMETHYLGALLIUM1445-79-0trimethylgallaneGallium, trimethyl-MFCD00014841Gallium trimethylEINECS 215-897-6Trimethylgallium, elec.gr.Trimethylgallium, elec. gr.(CH3)3GaDTXSID2061696AKOS015914830FT-0659225A808237Q419426Trimethylgallium, packaged for use in deposition systemsMolecular FormulaC3H9GaMolecular Weight114.83InChIInChI=1S/3CH3.Ga/h3*1H3InChI KeyXCZXGTMEAKBVPV-UHFFFAOYSA-NIsomeric SMILESC(C)C
Physical Data
No data available
Density, g·cm-3Measurement Temperature, °C1.453-130.161.448-153.161.453-140.16
Description (Association (MCS))Partner (Association (MCS))AdsorptionsilicaAdsorptionInSb(100)AdsorptionGaN(0001)
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Chemical shifts1Hbenzene-d6Chemical shifts13Cbenzene-d6Chemical shifts1H(2)H8-tolueneChemical shifts13C(2)H8-toluene
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Temperature (IR Spectroscopy), °CSpectrumneat (no solvent, gas phase)300.84Bandsneat (no solvent, gas phase)300.84Bandsfurther solvent(s)-153.16 - -133.16Bandsneat (no solvent)9.84
Description (UV/VIS Spectroscopy)Solvent (UV/VIS Spectroscopy)Comment (UV/VIS Spectroscopy)Spectrumgas190 nm - 310 nmSpectrum, Band assignmentneat (no solvent)106 nm - 270 nmSpectrumgaseous matrix190 nm - 390 nm
Route of Synthesis (ROS)
Route of Synthesis (ROS) of TRIMETHYLGALLIUM CAS 1445-79-0
ConditionsYieldWith magnesium In neat (no solvent) byproducts: MgI2; inert atm.; heating (autoclave, 120-160°C, 12 h); distn.;65%
Safety and Hazards
Pictogram(s)SignalDangerGHS Hazard StatementsH225 (15.64%): Highly Flammable liquid and vapor H250 (100%): Catches fire spontaneously if exposed to air H260 (84.36%): In contact with water releases flammable gases which may ignite spontaneously H261 (15.64%): In contact with water releases flammable gas H314 (100%): Causes severe skin burns and eye damage H318 (60.91%): Causes serious eye damage Precautionary Statement CodesP210, P222, P223, P231, P231+P232, P233, P240, P241, P242, P243, P260, P264, P264+P265, P280, P301+P330+P331, P302+P335+P334, P302+P361+P354, P303+P361+P353, P304+P340, P305+P354+P338, P316, P317, P321, P363, P370+P378, P402+P404, P403+P235, P405, and P501(The corresponding statement to each P-code can be found at the GHS Classification page.)
Other Data
Shelf Life1 year
DruglikenessLipinski rules componentMolecular Weight114.827logP1.584HBA0HBD0Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)0Rotatable Bond (RotB)0Matching Veber Rules2
Toxicity/Safety PharmacologyQuantitative Results
Use PatternTRIMETHYLGALLIUM CAS#: 1445-79-0 serves as a key precursor in thin film deposition technologies, including CVD and ALD. It is used to deposit high-quality gallium compound films in the semiconductor industry and other fields. And Trimethylgallium plays a critical role in the fabrication of optoelectronic devices, including solar cells and other optoelectronic devices.Trimethylgallium is also utilized in metalorganic chemistry as an organometallic precursor, participating in some organic synthesis reactions.
https://www.chemwhat.com/trimethylgallium-cas-1445-79-0/
Comments
Post a Comment