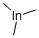
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
Product NameTRIMETHYLINDIUMIUPAC Nametrimethylindigane Molecular StructureCAS Registry Number 3385-78-2MDL NumberMFCD00014846Synonyms Trimethylindiumtrimethylindigane3385-78-2Indium, trimethyl-WR722GMA7HMFCD00014846EINECS 222-200-9UNII-WR722GMA7HTrimethylindium, elec. gr.DTXSID90187490IBEFSUTVZWZJEL-UHFFFAOYSA-NAKOS015914831Trimethylindium, elec gr. (99.999%-In)CS-0526987FT-0656820(3beta)-Cholest-5-en-3-ol3,4-dichlorobenzoateA821962Q4463086Molecular FormulaC3H9In Molecular Weight159.92InChIInChI=1S/3CH3.In/h3*1H3InChI KeyIBEFSUTVZWZJEL-UHFFFAOYSA-NIsomeric SMILESC(C)C
Patent InformationPatent IDTitlePublication DateCN111116618Process for preparing alkyl metal compounds]2020 US9884763Process for group III-V semiconductor nanostructure synthesis and compositions made using same2018WO2014/78263METHODS OF PRODUCING TRIMETHYLGALLIUM2014
Physical Data
No data available
Boiling Point, °C101 - 103140
Density, g·cm-3Reference Temperature, °CMeasurement Temperature, °C1.899-0.11.568191.5681919
Description (Association (MCS))Solvent (Association (MCS))AdsorptionTiO2 nanoparticles filmAdsorptionInSb(100)AdsorptionGaAsAdsorptionSi
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Spectrum1Hchloroform-d1Spectrum13Cchloroform-d1Chemical shifts1Hbenzene-d6Chemical shifts13Cbenzene-d6
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Temperature (IR Spectroscopy), °CSpectrumsolid matrix-259.16Bandsneat (no solvent)BandsgasBandssolid matrix
Route of Synthesis (ROS)
Route of Synthesis (ROS) of TRIMETHYLINDIUM CAS 3385-78-2
ConditionsYieldWith methylene chloride; sodium at 30 - 40℃; under 375.038 Torr; Flow reactor; Inert atmosphere;93%
Safety and Hazards
Pictogram(s)SignalDangerGHS Hazard StatementsH250 (100%): Catches fire spontaneously if exposed to air H260 (49.18%): In contact with water releases flammable gases which may ignite spontaneously H261 (20.77%): In contact with water releases flammable gas H314 (100%): Causes severe skin burns and eye damage H318 (60.66%): Causes serious eye damage Precautionary Statement CodesP210, P222, P223, P231, P231+P232, P233, P260, P264, P264+P265, P280, P301+P330+P331, P302+P335+P334, P302+P361+P354, P304+P340, P305+P354+P338, P316, P317, P321, P363, P370+P378, P402+P404, P405, and P501(The corresponding statement to each P-code can be found at the GHS Classification page.)
Other Data
No data available
DruglikenessLipinski rules componentMolecular Weight159.924logP1.584HBA0HBD0Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)0Rotatable Bond (RotB)0Matching Veber Rules2
Toxicity/Safety PharmacologyQuantitative Results
Use PatternTRIMETHYLINDIUM CAS#: 3385-78-2 can be utilized in CVD and ALD processes for the preparation of various indium compound films. And Trimethylindium is also employed in metalorganic chemistry as an organometallic precursor, participating in some organic synthesis reactions.Thin films of indium compounds deposited from trimethylindium are widely used in optoelectronics. This includes photovoltaic devices, laser devices, and other optical components.
https://www.chemwhat.com/trimethylindium-cas-3385-78-2/
Comments
Post a Comment