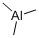
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
Product NameTrimethylaluminiumIUPAC Nametrimethylalumane Molecular StructureCAS Registry Number 75-24-1MDL NumberMFCD00008252SynonymsTrimethylaluminum75-24-1TRIMETHYLALUMINIUMAluminum, trimethyl-trimethylalumaneTrimethylalane(CH3)3AlAV210LG46JTrimethylaluminum, elec. gr.ALUMINUM TRIMETHYLMFCD00008252trimethyl aluminumAluminum trimethanideC3H9AlEINECS 200-853-0BRN 3587197UNII-AV210LG46JtrimethyaluminumAlMe3Me3Altri-methyl aluminiumTrimethylaluminum, 97%Al(Me)3EC 200-853-0Al(CH3)34-04-00-04397 (Beilstein Handbook Reference)TRIMETHYLALUMINUM Trimethylaluminum 2M in HexanesTrimethylaluminum 2M in TolueneDTXSID9058787Trimethylaluminum, 1M in heptaneTrimethylaluminum, 2M in heptaneTrimethylaluminum, 2M in tolueneAKOS015915463TRIMETHYLALUMINUM (AL(CH3)3)Trimethylaluminum, 25% w/w in hexaneUN 1103FT-0659349T0782T1575A838359Q416321Trimethylaluminum, packaged for use in deposition systemsTrimethylaluminum, electronic grade, >=99.9999% (ultra pure)Trimethylaluminum, elec.gr.;(99.999+%-Al), in 50 ml electropolished Swagelok cylinder (96-1077) for CVD/ALDMolecular FormulaC3H9AlMolecular Weight72.09InChIInChI=1S/3CH3.Al/h3*1H3InChI KeyJLTRXTDYQLMHGR-UHFFFAOYSA-NIsomeric SMILESC(C)C
Physical Data
No data available
Melting Point, °C 1515.415.114.5 - 1515015.2
Boiling Point, °CPressure (Boiling Point), Torr123.5 - 1258012548 - 5220129 - 13066 - 68100126760
Density, g·cm-3Reference Temperature, °CMeasurement Temperature, °C0.7478250.877-100.752420
Description (Association (MCS))Partner (Association (MCS))AdsorptionAgNO3TiCl4 activated MgCl2 filmAdsorptionInSb(100)
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzChemical shifts, Spectrum1Htetrahydrofuran-d8, toluene400Chemical shifts, Spectrum1Htetrahydrofuran-d8, hexaneChemical shifts, Spectrum1Hbenzene-d6, hexaneChemical shifts, Spectrum1Hbenzene-d626.84400.1
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Comment (IR Spectroscopy)Spectrumtoluene 2800 cm**-1 - 3700 cm**-1Bandstoluene2800 cm**-1 - 3700 cm**-1Spectrumsolid matrix400 cm**-1 - 4000 cm**-1Bandssolid matrix400 cm**-1 - 4000 cm**-1Spectrumfurther solvent(s)400 cm**-1 - 4000 cm**-1
Description (UV/VIS Spectroscopy)Solvent (UV/VIS Spectroscopy)Comment (UV/VIS Spectroscopy)Spectrumgas190 nm - 310 nm
Route of Synthesis (ROS)
Route of Synthesis (ROS) of Trimethylaluminium CAS#: 75-24-1
ConditionsYieldWith (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; potassium carbonate In tetrahydrofuran at 60℃; for 4h; Inert atmosphere;98%With palladium diacetate; tris-(o-tolyl)phosphine In tetrahydrofuran at 20℃; for 3h; Inert atmosphere; regioselective reaction;89%With bis-triphenylphosphine-palladium(II) chloride; potassium carbonate; triphenylphosphine In tetrahydrofuran at 60℃; for 3h; Inert atmosphere; regioselective reaction;83%With bis-triphenylphosphine-palladium(II) chloride; potassium carbonate; triphenylphosphine In tetrahydrofuran at 60℃; for 3h; Inert atmosphere;Experimental ProcedureGeneral procedure: Under a dry argon atmosphere, a mixture of Pd(PPh3)2Cl2 (0.0035 g,0.005 mmol), PPh3 (0.0026 g, 0.0100 mmol), and K2CO3 (0.138 g, 1.0mmol) were stirred in THF (2 mL). Then, AlMe3 (0.6 mmol) and propargylacetate (0.50 mmol) were added, respectively. The mixture was stirred for 3-4 h at 60 °C. After completion of the reaction, sat. aqNH4Cl (5 mL) was added and extracted with EtOAc (3 × 15 mL). The combined EtOAc layers was dried (Na2SO4), filtered, and evaporated under vacuum. The crude product was chromatographed on silica gel(hexane or EtOAc and hexane) to afford the corresponding allene product 2 or 5.83%With bis(triphenylphosphine)nickel(II) chloride; potassium carbonate; triphenylphosphine In tetrahydrofuran at 60℃; for 6h; Inert atmosphere;83%
Safety and Hazards
Pictogram(s)SignalDangerGHS Hazard StatementsH250 (98.72%): Catches fire spontaneously if exposed to air H260 (98.4%): In contact with water releases flammable gases which may ignite spontaneously H314 (98.72%): Causes severe skin burns and eye damage H318 (34.29%): Causes serious eye damage Precautionary Statement CodesP210, P222, P223, P231, P231+P232, P233, P260, P264, P264+P265, P280, P301+P330+P331, P302+P335+P334, P302+P361+P354, P304+P340, P305+P354+P338, P316, P317, P321, P363, P370+P378, P402+P404, P405, and P501(The corresponding statement to each P-code can be found at the GHS Classification page.)
Other Data
Shelf Life1 yearMarket Price
DruglikenessLipinski rules componentMolecular Weight72.086logP1.584HBA0HBD0Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)0Rotatable Bond (RotB)0Matching Veber Rules2
Use PatternTrimethylaluminium CAS#: 75-24-1 serves as a precursor material in the MOCVD method, used for depositing metal oxide films such as aluminum oxide. And In some organic synthesis reactions, Trimethylaluminium is used as a precursor for catalysts, playing a role in catalyzing certain reactions.
https://www.chemwhat.com/trimethylaluminium-cas-75-24-1/
Comments
Post a Comment