IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
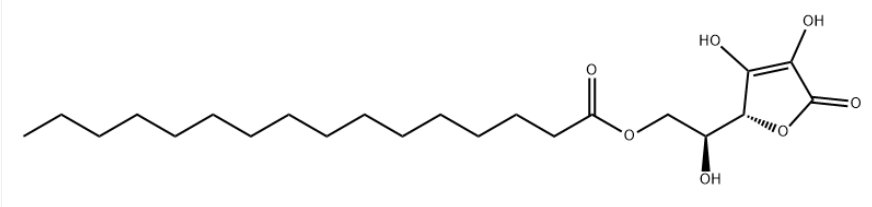
Product NameAscorbyl PalmitateIUPAC Name-2-hydroxyethyl] hexadecanoate Molecular StructureCAS Registry Number 137-66-6EINECS Number205-305-4MDL NumberMFCD00005377Beilstein Registry Number96552SynonymsASCORBYL PALMITATE137-66-6L-Ascorbyl 6-palmitate6-O-Palmitoyl-L-ascorbic acidL-Ascorbic acid 6-palmitateL-Ascorbic acid, 6-hexadecanoate(S)-2-((R)-3,4-Dihydroxy-5-oxo-2,5-dihydrofuran-2-yl)-2-hydroxyethyl palmitateL-Ascorbyl palmitateAscorbyl monopalmitateAscorbic palmitateCetyl ascorbate6-O-Palmitoylascorbic acidL-Ascorbic acid 6-hexadecanoateC22H38O76-O-Palmitoyl ascorbateIns no.304Ascorbyl palmitate Ascorbyl palmitate (VAN)UNII-QN83US2B0NIns-304Molecular FormulaC22H38O7Molecular Weight 414.5InChIInChI=1S/C22H38O7/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-18(24)28-16-17(23)21-19(25)20(26)22(27)29-21/h17,21,23,25-26H,2-16H2,1H3/t17-,21+/m0/s1 InChI KeyQAQJMLQRFWZOBN-LAUBAEHRSA-N Canonical SMILESCCCCCCCCCCCCCCCC(=O)OC(1C(=C(C(=O)O1)O)O)O
Physical Data
AppearanceWhite to light-yellow powder
Melting Point, °C Solvent (Melting Point) 113.1114.2170 - 174114 - 120112 - 114114 - 116116 - 117ethanol, petroleum ether
Description (Association (MCS))Solvent (Association (MCS))Temperature (Association (MCS)), °CPartner (Association (MCS))IR spectrum of the complex0 - 100L-DipalmitoylphosphatidylcholineFurther physical properties of the complexL-DipalmitoylphosphatidylcholineIR spectrum of the complex20 - 701,2-diperdeuteriopalmitoyl-sn-glycero-3-phosphocholineFurther physical properties of the complex1,2-diperdeuteriopalmitoyl-sn-glycero-3-phosphocholineIR spectrum of the complex0 - 1001,2-dipalmitoyl-3-sn-phosphatidylethanolamineIR spectrum of the complex20 - 701,2-dimyristoyl-sn-glycero-3-phosphoethanolamine
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzChemical shifts, Spectrum1Hdimethylsulfoxide-d6Chemical shifts1Hdimethylsulfoxide-d6300Chemical shifts13Cdimethylsulfoxide-d6Chemical shifts1Hacetone-d6300
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Temperature (IR Spectroscopy), °CBands, SpectrumBands, Spectrumpotassium bromideNear IR (NIR), Bands, Spectrum25SpectrumMid IR (MIR), BandsnujolBandsneat (no solvent)
Description (UV/VIS Spectroscopy)Solvent (UV/VIS Spectroscopy)Absorption Maxima (UV/VIS), nmSpectrum246SpectrumethanolAbsorption maximamethanol245Absorption maximamethanol240, 242
Route of Synthesis (ROS)
Route of Synthesis (ROS) of Ascorbyl Palmitate CAS 137-66-6
ConditionsYieldWith dmap at 80℃; for 8h; Temperature; Reagent/catalyst;99.45%Experimental Procedure Put palmitic anhydride (50g, 0.101mol), vitamin C (17.61g, 0.100mol) and DMAP (0.05g) into a 250mL four-necked flask with reflux and heating device, slowly raise the temperature to 80°C, react for 8 hours, and sample HPLC test showed that vitamin C residue ≤0.5% is qualified, stop heating, add n-hexane (200g) dropwise, drip in 0.5 hours, cool to 5-10, stir for 1 hour, filter, cool at 0-5 The filter cake was washed with hexane 3 times and dried to obtain the product vitamin C palmitate (41.29 g, 0.0994 mol), which was white crystals in appearance, HPLC purity: 99.84%, and molar yield: 99.45%.
Safety and Hazards
Pictogram(s)SignalWarningGHS Hazard StatementsH319 (46.15%): Causes serious eye irritation H412 (62.31%): Harmful to aquatic life with long lasting effects Precautionary Statement CodesP264+P265, P273, P280, P305+P351+P338, P337+P317, and P501(The corresponding statement to each P-code can be found at the GHS Classification page.)
Other Data
TransportationAt room temperature away from lightHS CodeStorageAt room temperature away from lightShelf Life2 yearsMarket Price
DruglikenessLipinski rules componentMolecular Weight414.54logP8.212HBA7HBD3Matching Lipinski Rules3Veber rules componentPolar Surface Area (PSA)113.29Rotatable Bond (RotB)18Matching Veber Rules1
Quantitative Results1 of 150Comment (Pharmacological Data)Bioactivities presentReferenceInhibition of hair growth2 of 150Comment (Pharmacological Data)Bioactivities presentReferenceSTABLE LIQUID SUSPENSION FORMULATION COMPRISING SYNTHETIC STEROIDS AND PROCESS FOR PRODUCING THE SAME 3 of 150Comment (Pharmacological Data)scriptionBioactivities presentReferencePHARMACEUTICAL DELIVERY SYSTEMS FOR HYDROPHOBIC DRUGS AND COMPOSITIONS COMPRISING SAME4 of 150Comment (Pharmacological Data)scriptionBioactivities presentReferencePersonal care compositions5 of 150Comment (Pharmacological Data)scriptionBioactivities presentReferenceMeasurement of superoxide dismutase-like activity of natural antioxidants.6 of 150Comment (Pharmacological Data)scriptionBioactivities presentReferenceEvaluation of peppermint oil and ascorbyl palmitate as inhibitors of cytochrome P4503A4 activity in vitro and in vivo7 of 150Comment (Pharmacological Data)scriptionBioactivities presentReferenceLipid-improving agent and composition containing lipid-improving agent8 of 150Comment (Pharmacological Data)scriptionBioactivities presentReferenceBiosynthesis of ascorbyl benzoate in organic solvents and study of its antioxygenic and antimicrobial properties
Use PatternAscorbyl Palmitate CAS#: 137-66-6 is an efficient, safe, and non-toxic liposoluble nutritional antioxidant that is insoluble in water and vegetable oil. It appears as a white powder with a slight citrus odor.
https://www.chemwhat.com/ascorbyl-palmitate-cas-137-66-6/
Comments
Post a Comment