IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
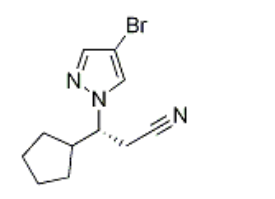
Product Name(R)-3-(4-broMo-1H-pyrazol-1-yl)-3-cyclopentylpropanenitrileIUPAC Name(3R)-3-(4-bromopyrazol-1-yl)-3-cyclopentylpropanenitrile Molecular StructureCAS Registry Number 1146629-83-5MDL NumberMFCD16038969Synonyms1146629-83-5(R)-3-(4-Bromo-1H-pyrazol-1-yl)-3-cyclopentylpropanenitrile(3R)-3-(4-bromo-1H-pyrazol-1-yl)-3-cyclopentylpropanenitrile1H-Pyrazole-1-propanenitrile, 4-bromo-beta-cyclopentyl-, (betaR)-1H-Pyrazole-1-propanenitrile, 4-bromo-b-cyclopentyl-, (bR)-SCHEMBL3004297DTXSID40738416CS-B0071MFCD16038969F869911H-Pyrazole-1-propanenitrile,4-bromo-b-cyclopentyl-,(bR)-Molecular FormulaC11H14BrN3Molecular Weight268.15InChIInChI=1S/C11H14BrN3/c12-10-7-14-15(8-10)11(5-6-13)9-3-1-2-4-9/h7-9,11H,1-5H2/t11-/m1/s1 InChI KeyXXUIJTAHLDUGJF-LLVKDONJSA-N Canonical SMILESC1CCC(C1)(CC#N)N2C=C(C=N2)Br
Physical Data
AppearanceOff-white solid
Melting Point, °C Solvent (Melting Point) 82 - 83cyclohexane
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzChemical shifts1Hdimethylsulfoxide-d6Chemical shifts1Hchloroform-d1Chemical shifts1Hchloroform-d1Chemical shifts1Hdimethylsulfoxide-d6
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Mid IR (MIR), Bandspotassium bromide
Route of Synthesis (ROS)
Route of Synthesis (ROS) of (R)-3-(4-broMo-1H-pyrazol-1-yl)-3-cyclopentylpropanenitrile CAS#: 1146629-83-5
ConditionsYieldWith phosphorous pentoxide In tetrahydrofuran at 70℃; for 2h; Inert atmosphere;98%With trichlorophosphate In tetrahydrofuran at 70℃; for 2h; Reagent/catalyst; Inert atmosphere;98%With hydrogen; triethylamine In ethanol; water at 110℃; under 30003 Torr; for 24h; Autoclave;98%With trichlorophosphate In tetrahydrofuran at 70℃; for 2h; Reagent/catalyst; Inert atmosphere;Experimental ProcedureTo a solution of 17 (1.24 g, 4.3 mol, 1.0 eq.), in tetrahydrofuran (50 mL), added phosphorus pentoxide (1.84 g, 13.0 mol, 3 eq.) under argon. The reaction mixture was stirred for 2 hours at 70 °C, diluted with ethyl acetate (200 mL), and quenched by adding saturated sodium bicarbonate (200 mL). The aqueous layer was back-extracted with ethyl acetate (2 x 100 mL). The organic phases were combined, washed with water, brine, dried over anhydrous Na2SO4 and filtered. The filtrate was concentrated under reduced pressure and purified by SiO2 chromatography using methanol/dichloromethane (5 %) as eluent to provide the title compound as a white solid (1.13 g, 98%). HPLC 100% (tR = 12.56 min, CH3OH in 0.1% TFA water 5%~95% in 20 min); 1 H NMR (500 MHz, DMSO-d6) δ 8.12 (d, J= 0.5 Hz, 1H), 7.63 (d, J = 0.5 Hz, 1H), 4.38 (td, J = 9.4, 4.7 Hz, 1H), 3.15 - 3.08 (m, 2H), 2.33 - 2.28 (m, 1H), 1.79 - 1.72 (m, 1H), 1.61 - 1.38 (m, 4H), 1.30 - 1.05 (m, 3H); HPLC-MS (ESI+): m/z 268.0 (M+H)+.98%
Safety and Hazards
No data available
Other Data
TransportationStore at room temperature, sealed and away from lightHS CodeStorageStore at room temperature, sealed and away from lightShelf Life2 yearsMarket Price
Use Pattern(R)-3-(4-broMo-1H-pyrazol-1-yl)-3-cyclopentylpropanenitrile CAS#: 1146629-83-5 is used in pharmaceutical intermediates.
https://www.chemwhat.com/r-3-4-bromo-1h-pyrazol-1-yl-3-cyclopentylpropanenitrile-cas-1146629-83-5-3/
Comments
Post a Comment