IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
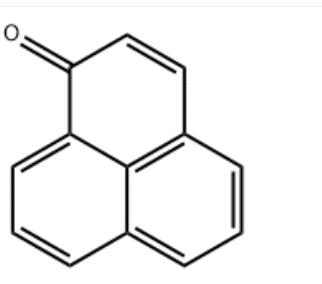
Product NamePerinaphthenone IUPAC Namephenalen-1-one Molecular StructureCAS Registry Number 548-39-0EINECS Number208-945-2MDL NumberMFCD00004143SynonymsPhenalen-1-onePERINAPHTHENONE1H-Phenalen-1-one548-39-0Phenalenone7-PerinaphthenoneCCRIS 4923EINECS 208-945-2EVU6EX9G8HNSC 150161NSC-150161PerinaphthenonPhenalenone-1-onePerinaphthenone, 97%starbld0009638UNII-EVU6EX9G8HSCHEMBL188491CCRIS-4923CHEMBL2275111H-BENZONAPHTHEN-1-ONEWWBGWPHHLRSTFI-UHFFFAOYSA-DTXSID20203278MFCD00004143NSC150161AKOS004902220AB92503AS-82921LS-102578CS-0331802FT-0632364D79953AE-562/43459279J-504714Molecular FormulaC13H8OMolecular Weight180.2InChIInChI=1S/C13H8O/c14-12-8-7-10-4-1-3-9-5-2-6-11(12)13(9)10/h1-8H InChI KeyWWBGWPHHLRSTFI-UHFFFAOYSA-N Canonical SMILESC1=CC2=C3C(=C1)C=CC(=O)C3=CC=C2
Physical Data
AppearanceYellow solid
Melting Point, °C Solvent (Melting Point) 144 - 145155 - 157148 - 149149 - 151155 - 156benzene
Description (Association (MCS))Solvent (Association (MCS))Temperature (Association (MCS)), °CPartner (Association (MCS))Further physical properties of the adsorbed moleculequartzUV/VIS spectrum of the complexCDCl3nile blue A perchlorate, O2Stability constant of the complex with ...H2Oβ-cyclodextrinStability constant of the complex with ...H2Oβ-hydroxypropyl-cyclodextrin, molar substitution 0.85
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzChemical shifts, Spectrum1Hchloroform-d1400Chemical shifts13Cchloroform-d1100Chemical shifts1Hchloroform-d1400Chemical shifts1Hchloroform-d1400Chemical shifts13Cchloroform-d1100
Description (IR Spectroscopy)Solvent (IR Spectroscopy)BandsCD3CNBandsCHCl3BandsKBrSpectrumgas
Description (UV/VIS Spectroscopy)Solvent (UV/VIS Spectroscopy)Absorption Maxima (UV/VIS), nmSpectrumSpectrumacetonitrileSpectrumaq. phosphate bufferSpectrummethanolSpectrummethanol204, 248, 361
Route of Synthesis (ROS)
Route of Synthesis (ROS) of Perinaphthenone CAS 548-39-0
ConditionsYieldStage #1: naphthalene With aluminum (III) chloride In dichloromethane for 0.0833333h; Cooling with ice;Stage #2: cinnamoyl chloride In dichloromethane at 20℃; for 0.833333h;76%With carbon disulfide; aluminium trichlorideExperimental ProcedureNaphthalene (0.64 g, 5 mmol) and anhydrous aluminum chloride (1.4 g, 5.5 mmol) were dissolved in dichloromethane (10 mL) and stirred in an ice bath for 5 min, then solution of cinnamoyl chloride (0.83 g, 5 mmol) in 2.5 ml dichloromethane was added dropwise to the flask during 30 min and reacted at room temperature for 20 min. After that, the reaction solution was rotary evaporated under vacuum condition and purified with silica gel chromatography using petroleum ether/ethyl acetate (10: 1) as eluting solvent to afford phenalenone as a yellow solid with a yield of 76% (0.40 g). m.p. 144-145 oC. 1H NMR (400 MHz, Chloroform-d) δ 8.59 (d, J = 7.6 Hz, 1H), 8.16 (d, J = 8.0 Hz, 1H), 7.98 (d, J = 8.0 Hz, 1H), 7.77 - 7.67 (m, 3H), 7.55 (t, J = 7.6 Hz, 1H), 6.71 (d, J = 9.8 Hz, 1H). 13C NMR (100 MHz, Chloroform-d) δ 185.14, 137.17, 132.90, 132.63, 131.84, 131.61, 131.51, 130.24, 129.30, 129.25, 127.89, 127.15, 126.61. HRMS (ESI): m/z calcd. for C13H9O+ +: 181.0653; found: 181.0654
Safety and Hazards
Pictogram(s)SignalWarningGHS Hazard StatementsH302 (50%): Harmful if swallowed H315 (50%): Causes skin irritation H319 (50%): Causes serious eye irritation H335 (50%): May cause respiratory irritation Precautionary Statement CodesP261, P264, P264+P265, P270, P271, P280, P301+P317, P302+P352, P304+P340, P305+P351+P338, P319, P321, P330, P332+P317, P337+P317, P362+P364, P403+P233, P405, and P501(The corresponding statement to each P-code can be found at the GHS Classification page.)
Other Data
TransportationRoom Temperature and Long-term storage 2-8°CHS CodeStorageRoom Temperature and Long-term storage 2-8°CShelf Life1 yearMarket Price
DruglikenessLipinski rules componentMolecular Weight180.206logP3.092HBA1HBD0Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)17.07Rotatable Bond (RotB)0Matching Veber Rules2
Quantitative Results1 of 44Comment (Pharmacological Data)Bioactivities presentReferenceFluorescent haloalkyl derivatives of reporter molecules well retained in cells2 of 44Comment (Pharmacological Data)Bioactivities presentReferenceHydrolyzable fluorescent substrates and analytical determinations using same3 of 44Comment (Pharmacological Data)Bioactivities presentReference4 of 44Comment (Pharmacological Data)Bioactivities presentReference
Use PatternPerinaphthenone CAS#: 548-39-0 as an important intermediate in the synthesis of various pharmaceuticals, including anticancer drugs, antibiotics, and antimalarial drugs. And it is extensively employed in photosensitive materials due to its ability to absorb ultraviolet and visible light. Perinaphthenone is used in the production of organic semiconductor materials and is utilized as an antioxidant and preservative in cosmetics and personal care products.
https://www.chemwhat.com/perinaphthenone-cas-548-39-0-2/
Comments
Post a Comment