IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
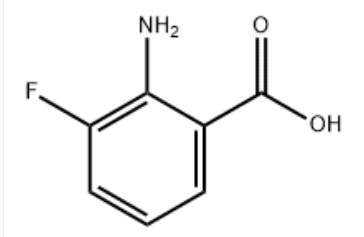
Product Name3-Fluoroanthranilic AcidineIUPAC Name2-amino-3-fluorobenzoic acid Molecular StructureCAS Registry Number 825-22-9EINECS Number630-115-7MDL NumberMFCD01569395Beilstein Registry NumberSynonyms2-Amino-3-fluorobenzoic acid825-22-93-Fluoroanthranilic Acid2-amino-3-fluoro-benzoic acid2-amino-3-fluorobenzenecarboxylic acidBenzoic acid, 2-amino-3-fluoro-2-Azanyl-3-Fluoranyl-Benzoic AcidMFCD01569395C7H6FNO24owm3-fluoroanthranilateSCHEMBL1619512-Amino-3-fluorobenzoic acid #DTXSID70342984ACT00442ACT00505BCP24579ZINC2574870BBL100542STL554336AKOS005073124AB09241AC-3880CS-W002787KC-0713SY0052782-Amino-3-fluorobenzoic acid, AldrichCPRDB-024255AM20060112F0570FT-0602026EN300-56009825A229Q-103166Q27453765Z362758748Molecular FormulaC7H6FNO2Molecular Weight155.126InChIInChI=1S/C7H6FNO2/c8-5-3-1-2-4(6(5)9)7(10)11/h1-3H,9H2,(H,10,11)InChI KeyC1=CC(=C(C(=C1)F)N)C(=O)OCanonical SMILESC1=CC(=CN=C1)N
Physical Data
AppearanceOff white to brown yellow crystalline powder
Melting Point, °C Solvent (Melting Point) 183 - 184CHCl3171177183aq. ethanol
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzChemical shifts1Hacetone500.1Chemical shifts13Cacetone125.6Spectrum1Hdimethylsulfoxide-d6Chemical shifts19Fdimethylsulfoxide-d6Chemical shifts19Facetone-d6
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Temperature (IR Spectroscopy), °CBandspotassium bromide2-Amino-3-fluorobenzoic acid:
Description (UV/VIS Spectroscopy)Solvent (UV/VIS Spectroscopy)Comment (UV/VIS Spectroscopy)Absorption Maxima (UV/VIS), nmExt./Abs. Coefficient, l·mol-1cm-1aq. phosphate buffer308
Route of Synthesis (ROS)
Route of Synthesis (ROS) of 3-Fluoroanthranilic Acid CAS-825-22-9
ConditionsYieldWith N-Bromosuccinimide In dichloromethane at 0 - 20℃; for 1h;82%With N-Bromosuccinimide In N,N-dimethyl-formamide at -10℃; for 1h; Inert atmosphere;80%With N-Bromosuccinimide In dichloromethane at 20℃; for 3h;80%Experimental Procedure Step a. NBS (114.8 g, 645 mmol) was added in a portion wise manner over 1 h to a stirred orange/brown suspension of 2-amino-3-fluorobenzoic acid (100 g, 645 mmol) in DCM (1000 ml) at rt, and the resulting mixture was stirred at rt for 2 h. The mixture was then filtered and the resulting solid was washed sequentially with DCM (2 x 250 ml) and water (3 x 400 ml) before being dried under vacuum at 55°C for 7 h to give the desired product as a beige solid (121.4 g, 80%). LCMS (Method S): rt 2.93 min, m/z 232/234 -; NMR (400 MHz, DMSO-d6) δ ppm 7.64 (m, 1H), 7.52 (dd, J= 10.9, 2.3 Hz, 1H)
Safety and Hazards
Pictogram(s)SignalWarningGHS Hazard StatementsH302 (25%): Harmful if swallowed H315 (75%): Causes skin irritation H319 (75%): Causes serious eye irritation H335 (100%): May cause respiratory irritation Precautionary Statement CodesP261, P264, P264+P265, P270, P271, P280, P301+P317, P302+P352, P304+P340, P305+P351+P338, P319, P321, P330, P332+P317, P337+P317, P362+P364, P403+P233, P405, and P501(The corresponding statement to each P-code can be found at the GHS Classification page.)ECHA C&L Notifications SummaryAggregated GH
Other Data
TransportationUnder the room temperature and away from lightHS CodeStorageUnder the room temperature and away from lightShelf Life2 yearsMarket Price
DruglikenessLipinski rules componentMolecular Weight155.129logP1.41HBA3HBD2Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)63.32Rotatable Bond (RotB)1Matching Veber Rules2
BioactivityQuantitative Results
Quantitative Results1 of 14Comment (Pharmacological Data)Bioactivities presentReference4-Hydroxy-3-quinoline-carboxylic acid derivatives2 of 14Comment (Pharmacological Data)Bioactivities presentReferenceADENOSINE A2A RECEPTOR ANTAGONISTS3 of 14Comment (Pharmacological Data)Bioactivities presentMeasurementDissociation constant4 of 10Assay DescriptionHydrogen bond acidity of the compound was determinedReferenceSubstituents effects on activity of kynureninase from Homo sapiens and Pseudomonas fluorescens
Use Pattern3-Fluoroanthranilic Acid CAS#: 825-22-9 is used as a pharmaceutical intermediate.
https://www.chemwhat.com/3-fluoroanthranilic-acid-cas-825-22-9/
Comments
Post a Comment