IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
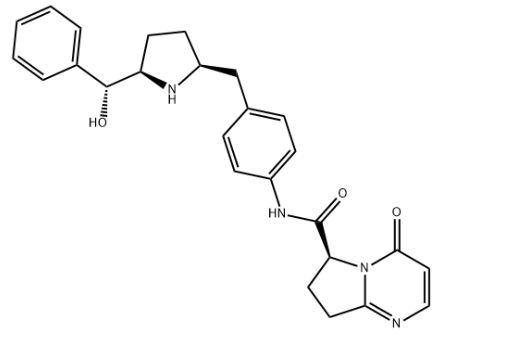
Product NameVibegronIUPAC Name(6S)-N-pyrrolidin-2-yl]methyl]phenyl]-4-oxo-7,8-dihydro-6H-pyrrolopyrimidine-6-carboxamide Molecular StructureCAS Registry Number 1190389-15-1SynonymsVIBEGRON1190389-15-1KRP-114VGemtesaMK-4618(S)-N-(4-(((2S,5R)-5-((R)-hydroxy(phenyl)methyl)pyrrolidin-2-yl)methyl)phenyl)-4-oxo-4,6,7,8-tetrahydropyrrolopyrimidine-6-carboxamideM5TSE03W5UC26H28N4O3Molecular FormulaC26H28N4O3Molecular Weight444.5InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 InChI KeyDJXRIQMCROIRCZ-XOEOCAAJSA-N
Patent InformationPatent IDTitlePublication DateUS2009/253705HYDROXYMETHYL PYRROLIDINES AS BETA 3 ADRENERGIC RECEPTOR AGONISTS2009
Physical Data
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzSpectrum1Hdimethylsulfoxide-d626.84400Chemical shifts, Spectrum1Hdimethylsulfoxide-d626.84500COSY (Correlation Spectroscopy), Spectrum1H, 1Hdimethylsulfoxide-d626.84500ROESY (Rotating frame Overhauser Enhancement Spectroscopy), Spectrum1H, 1Hdimethylsulfoxide-d626.84500HSQC (Heteronuclear Single Quantum Coherence), Spectrum1H, 13Cdimethylsulfoxide-d626.84HMBC (Heteronuclear Multiple Bond Coherence), Spectrum1H, 13Cdimethylsulfoxide-d626.84Spectrum13Cdimethylsulfoxide-d626.84126
Route of Synthesis (ROS)
Route of Synthesis (ROS) of vibegron-cas-1190389-15-1
ConditionsYieldStage #1: tert-butyl(2R,5S)-2--5-pyrimidin-6-yl]carbonyl}amino)benzyl]pyrrolidine-1-carboxylate With trifluoroacetic acid In dichloromethane at 20℃; for 1.5h;Stage #2: With sodium hydrogencarbonate pH=8 - 9;60%Stage #1: tert-butyl(2R,5S)-2--5-pyrimidin-6-yl]carbonyl}amino)benzyl]pyrrolidine-1-carboxylate With trifluoroacetic acid In dichloromethane at 20℃; for 1.5h;Stage #2: With sodium hydrogencarbonate pH=8 - 9;60%Experimental ProcedureTo a solution of the intermediate from Step A (2.50 g, 4.59 mmol) in dichloromethane (40 ml) was added trifluoroacetic acid (15 ml). The reaction mixture was stirred at ambient temperature for 1.5 h. After removal of the volatiles, saturated NaHCO3 was added to make the PH value to 8-9. The mixture was then extracted with dichloromethane. The combined organic layers were dried over Na2SO4. After concentration, crystallization from methanol/acetonitrile afforded the title compound as a white solid (1.23 g, 60%). 1H NMR (DMSO-d6): δ 10.40 (s, 1H), 7.91 (d, J=6.7 Hz, 1H), 7.49 (d, J=8.3 Hz, 2H), 7.32-7.26 (m, 4H), 7.21 (m, 1H), 7.15 (d, J=8.4 Hz, 2H), 6.23 (d, J=6.7 Hz, 1H), 5.11 (dd, J=9.6, 2.9 Hz, 1H), 5.10 (br, 1H), 4.21 (d, J=7.1 Hz, 1H), 3.20-3.00 (m, 4H), 2.66-2.51 (m, 3H), 2.16 (m, 1H), 1.57 (m, 1H), 1.38 (m, 1H), 1.29-1.23 (m, 2H). LC-MS 445.3 (M+1).
Safety and Hazards
No data available
Other Data
TransportationUnder the room temperature and away from lightHS CodeStorageUnder the room temperature and away from lightShelf Life1 yearMarket Price
DruglikenessLipinski rules componentMolecular Weight444.533logP1.581HBA7HBD3Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)94.03Rotatable Bond (RotB)7Matching Veber Rules2
Quantitative Results1 of 13Comment (Pharmacological Data)Bioactivities presentReferenceHYDROXYMETHYL PYRROLIDINES AS BETA 3 ADRENERGIC RECEPTOR AGONISTS2 of 13Comment (Pharmacological Data)Bioactivities presentReferenceAN AGENT FOR TREATING NOCTURNAL POLLAKIURIA3 of 13Comment (Pharmacological Data)Bioactivities presentReferencePollakiuria night therapeutic agent4 of 13Comment (Pharmacological Data)Bioactivities presentReferenceProcess for preparing beta 3 agonists and intermediates5 of 13 Comment (Pharmacological Data)Bioactivities presentReferenceAgent for treating nocturnal pollakiuria6 of 13Comment (Pharmacological Data)physiological behaviour discussedReferenceSelectivity and Maximum Response of Vibegron and Mirabegron for β3-Adrenergic Receptors7 of 13Comment (Pharmacological Data)physiological behaviour discussedReferenceMETHODS OF TREATING HEART FAILURE WITH VIBEGRON
Toxicity/Safety PharmacologyQuantitative Results
Use PatternVibegron CAS#: 1190389-15-1 is an intermediate in pesticides and dyes; pesticide raw materials; analytical reagents.
https://www.chemwhat.com/vibegron-cas-1190389-15-1/
Comments
Post a Comment