IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
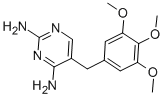
Product NameTrimethoprimIUPAC Name 5-pyrimidine-2,4-diamine Molecular StructureCAS Registry Number 738-70-5EINECS Number212-006-2MDL NumberMFCD00006400Beilstein Registry Number625127Synonymstrimethoprim738-70-5ProloprimTrimpexTrimetoprimBactraminMonotrimMonotriminTrimopanWellcoprim2,4-Diamino-5-(3,4,5-trimethoxybenzyl)pyrimidineMonoprimSyraprimTrimanylTriprimUretrim5-(3,4,5-trimethoxybenzyl)pyrimidine-2,4-diamineTrimethoprimumTrimethoprime5-pyrimidine-2,4-diamineNSC-106568NIH 204Primsolcomponent of BactrimBW 56-722,4-Pyrimidinediamine, 5--InfectotrimetTCMDC-1255385-(3,4,5-Trimethoxybenzyl)-2,4-pyrimidinediamineTrimethoprim-d95--2,4-pyrimidinediamineNSC 1065685-(3,4,5-Trimethoxybenzyl)-2,4-diaminopyrimidineBW-56-72CHEBI:45924CHEMBL22Trimpex (TN)2,4-Pyrimidinediamine, 5-((3,4,5-trimethoxyphenyl)methyl)-AbaprimMFCD00036761Apo-SulfatrimBW 5672Pyrimidine, 2,4-diamino-5-(3,4,5-trimethoxybenzyl)-MLS000079023AN164J8Y0XBriscotrimNovotrimelStreptoplusSulfoxaprimTrimethioprimUrobactrimWellcoprinAnitrimAntrimaAntrimoxBacidalBacticelBactoprimBencoleBethaprimBiosultenChemotrinColizoleConprimCotrimelDuocideEsbesulEspectrinEuctrimExbesulFermagexFortrimIkaprimKombinaxLagatrimLastrimMetoprimPancidimProtrinResprimSalvatrimSetprinSinotrimSugaprimSulfamarSulthrimSultrexTrimexolTrimezolTrimonoTrisulcomTrisulfamTrisuralUtetrinVelatenXeroprimZamboprim2,4-Pyrimidinediamine, 5-((3,4,5-trimethoxyphenyl)-methyl)-BacdanBacideDeprimOmstatPurbalRoubacRoubalStopanToprimTrisulBacinBactaFutinNIH-204Trimpex 200Co-TrimoxizoleLagatrim ForteSeptrin ForteAlcorim-FColizole DSSeptrin SSeptrin DSSmz-TmpNSC106568Trimez-IFSAU-Princomponent of SeptraNCGC00016055-05Trimethopriom5-((3,4,5-Trimethoxyphenyl)methyl)-2,4-pyrimidinediamineBactiforCAS-738-70-5DosulfinInstalacSMR000035999Trimetoprim TrimogalLescotTiempeTrimetoprim Resprim ForteTrimethoprim 100 microg/mL in AcetonitrileUro-D STmp Smx5-(3,4,5-Trimethoxy-benzyl)-pyrimidine-2,4-diamineTrimethoprime Trimethoprimum Trimetoprima 2,4-Diamino-5-(3',4',5'-trimethoxybenzyl)pyrimidineBacterial NIH 204 (VAN)Trimethoprim D3 (4-methoxy D3)Proloprim (TN)WR 5949CCRIS 2410HSDB 6781SR-01000075652EINECS 212-006-25-(3, 4, 5-Trimethoxybenzyl)-2, 4-pyrimidinediamineBRN 0625127UNII-AN164J8Y0XTrimethoprim (JAN/USP/INN)5-{methyl}pyrimidine-2,4-diamineAI3-52594Trimethoprim D3 (4-methoxy D3) 100 microg/mL in AcetonitrileB-LockKUC103659NTrimethoprim,(S)Prestwick_485KSC-4-158Trimethoprim (TMP)Bactrim (Salt/Mix)Spectrum_000167Tocris-0650Trimethoprim 2w9h3fl93n0h3s3v4km2Opera_ID_1760Prestwick0_000208Prestwick1_000208Prestwick2_000208Prestwick3_000208Spectrum2_000937Spectrum3_000643Spectrum4_000372Spectrum5_001559Lopac-T-7883TRIMETHOPRIM TRIMETHOPRIM TRIMETHOPRIM Epitope ID:119684UPCMLD-DP132T 7883TRIMETHOPRIM TRIMETHOPRIM TRIMETHOPRIM Lopac0_001271Oprea1_495058SCHEMBL24506BSPBio_000195BSPBio_002245KBioGR_000863KBioSS_000647TRIMETHOPRIM 5-25-13-00429 (Beilstein Handbook Reference)MLS001201740MLS002303068MLS002548881BIDD:GT0190DivK1c_000589SPECTRUM1500595TRIMETHOPRIM TRIMETHOPRIM SPBio_000874SPBio_002116BPBio1_000215DTXSID3023712UPCMLD-DP132:001BDBM18069GTPL10931HMS501N11KBio1_000589KBio2_000647KBio2_003215KBio2_005783KBio3_001465TRIMETHOPRIM Trimethoprim, >=98% (HPLC)NINDS_0005892,4,5-trimethoxybenzyl)pyrimidineHMS1568J17HMS1921I03HMS2090D14HMS2092A10HMS2095J17HMS2230L06HMS3259I11HMS3263P04HMS3371O18HMS3652E03HMS3712J17Pharmakon1600-01500595TRIMETHOPRIM TRIMETHOPRIM Trimethoprim for system suitabilityTRIMETHOPRIM 2,4,5-trimethoxyphenzyl)pyrimidineALBB-028968BCP12148COTRIM COMPONENT TRIMETHOPRIMHY-B0510SEPTRA COMPONENT TRIMETHOPRIMZINC6627681Co-trimoxazole component trimethoprimTox21_110291Tox21_200157Tox21_501271TRIMETHOPRIM BACTRIM COMPONENT TRIMETHOPRIMBBL005584CCG-40335NSC752719NSC757370s3129STK177322STL455117UROPLUS COMPONENT TRIMETHOPRIMTRIMETHOPRIMUM AKOS001650069SULFATRIM COMPONENT TRIMETHOPRIMSULMEPRIM COMPONENT TRIMETHOPRIMTox21_110291_1AC-8427BW-5672DB00440KS-1145LP01271NC00483NSC-752719NSC-757370SDCCGSBI-0051237.P004TRIMETHOPRIM COMPONENT OF COTRIMTRIMETHOPRIM COMPONENT OF SEPTRABACTRIM DS COMPONENT TRIMETHOPRIMIDI1_000589SMP2_000262TRIMETHOPRIM COMPONENT OF BACTRIMTRIMETHOPRIM COMPONENT OF UROPLUSNCGC00016055-01NCGC00016055-02NCGC00016055-03NCGC00016055-04NCGC00016055-06NCGC00016055-07NCGC00016055-08NCGC00016055-09NCGC00016055-10NCGC00016055-11NCGC00016055-12NCGC00016055-13NCGC00016055-14NCGC00016055-16NCGC00016055-17NCGC00016055-27NCGC00024707-01NCGC00024707-03NCGC00024707-04NCGC00024707-05NCGC00024707-06NCGC00024707-07NCGC00024707-08NCGC00257711-01NCGC00261956-01COTRIM D.S. COMPONENT TRIMETHOPRIMSY031734TRIMETHOPRIM COMPONENT OF SULFATRIMTRIMETHOPRIM COMPONENT OF SULMEPRIMTrimethoprim/sulfamethoxazole (commercial)SBI-0051237.P003DB-055812SULFAMETHOPRIM COMPONENT TRIMETHOPRIMTRIMETHOPRIM COMPONENT OF BACTRIM DS2, 5--AB00052118BB 0258034EU-0101271FT-0601630FT-0675578FT-0675579FT-0675580SW196690-3T2286Trimethoprim 1000 microg/mL in AcetonitrileTRIMETHOPRIM COMPONENT OF COTRIM D.S.BACTRIM PEDIATRIC COMPONENT TRIMETHOPRIMC01965D00145EN300-118703TRIMETHOPRIM COMPONENT OF SULFAMETHOPRIMTrimethoprim, crystallized, >=99.0% (HPLC)WLN: T6N CNJ BZ DZ E1R CO1 DO1 EO15-(3,5-Trimethoxybenzyl)-2,4-diaminopyrimidineAB00052118-30AB00052118-32AB00052118_33AB00052118_34Trimethoprim, VETRANAL(TM), analytical standard738T705Q422665TRIMETHOPRIM COMPONENT OF BACTRIM PEDIATRIC2,4-diamino-5-(3,4,5-trimethoxybenzyl) pyrimidine2,4-diamino-5-(3,4,5-trimethoxybenzyl)-pyrimidinePyrimidine,4-diamino-5-(3,4,5-trimethoxybenzyl)-SR-01000075652-1SR-01000075652-3SR-01000075652-6W-1044415-(3,4,5-Trimethoxybenzyl)-2,4-pyrimidinediamine #BRD-K07208025-001-06-5SR-01000075652-102-amino-5-(3,4,5-trimethoxybenzyl)-4-pyrimidinylamineF0914-5266Trimethoprim, certified reference material, TraceCERT(R)Z15153850715-(3,4,5-trimethoxybenzyl)pyrimidine-2,4(1H,3H)-diimineTrimethoprim, British Pharmacopoeia (BP) Reference StandardTrimethoprim, European Pharmacopoeia (EP) Reference StandardTrimethoprim, United States Pharmacopeia (USP) Reference StandardTrimethoprim for system suitability, European Pharmacopoeia (EP) Reference StandardTrimethoprim, Pharmaceutical Secondary Standard; Certified Reference MaterialMolecular FormulaC14H18N4O3 Molecular Weight290.32InChIInChI=1S/C14H18N4O3/c1-19-10-5-8(6-11(20-2)12(10)21-3)4-9-7-17-14(16)18-13(9)15/h5-7H,4H2,1-3H3,(H4,15,16,17,18) InChI KeyIEDVJHCEMCRBQM-UHFFFAOYSA-NCanonical SMILESCOC1=CC(=CC(=C1OC)OC)CC2=CN=C(N=C2N)N
Patent InformationPatent IDTitlePublication DateWO2017/29202HYPERPHENYLALANINEMIA AND TREATMENTS THEREOF2017US39857453-Imino-1,2,4-benzotriazine-1-oxides1976US4115650Process for preparing 2,4-diamino-5-(substituted benzyl)-pyrimidines1978
Physical Data
AppearanceWhite Specifications or yellowish white powder
Melting Point, °C 199 - 203201.8138 - 139200 - 203
Density, g·cm-3Reference Temperature, °C1.329-173.161.24
Description (Association (MCS))Solvent (Association (MCS))Temperature (Association (MCS)), °CPartner (Association (MCS))Association with compound17.84bovine serum albuminAssociation with compound17.84human serum albuminAssociation with compoundrandomly methylated β-cyclodextrinAssociation with compoundethanol, H2O24.85 - 44.85montmorillonite K10
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzSpectrum1Hwater-d2Chemical shifts1Hdimethylsulfoxide-d6Chemical shifts, Spectrum1Hdimethylsulfoxide-d6300
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Temperature (IR Spectroscopy), °CBands, Spectrum ATR (attenuated total reflectance), SpectrumBandspotassium bromide
Description (UV/VIS Spectroscopy)Solvent (UV/VIS Spectroscopy)Comment (UV/VIS Spectroscopy)Absorption Maxima (UV/VIS), nmSpectrumwaterSpectrumdichloromethaneRatio of solvents: 0.1NSpectrumaq. phosphate buffer270.6
Route of Synthesis (ROS)
Route of Synthesis (ROS) of trimethoprim-cas-738-70-5-2
ConditionsYieldWith tin(IV) chloride In chloroform at 20℃90.58%With tin(IV) chloride In chloroform for 1h; Solvent; Reflux90.58%With tin(IV) chloride In chloroform for 1h; Reflux90.58%Experimental Procedure8 Example 8 Preparation of compound 2 (N, N '-(5- (2-acetyl-3,4,5-trimethoxybenzyl) pyrimidine-2,4-diyl) diacetamide)Add Compound 1 (trimethoprim) (10.00g, 34.45mmol) to a 250ml reaction flask,Acetyl chloride (9.80 ml, 138.57 mmol) and 100 ml of chloroform, and tin tetrachloride (8.00 ml, 68.36 mmol) was added under stirring, and the reaction was refluxed for 1 h.Cool down to room temperature, pour the reaction solution into 50ml ice water, stir for 6mins, separate the liquid, wash the organic phase 3 times with 5ml water,Combine the aqueous phases, extract the aqueous phase 3 times with 5 ml of chloroform, combine the organic phases, adjust the pH of the saturated sodium carbonate aqueous solution to 7-8, and separate the liquid organic phase 5 times with the aqueous phase.It was dried over anhydrous sodium sulfate, filtered, and concentrated in vacuo. The ethylene glycol monomethyl ether was recrystallized to obtain 12.98 g of the product with a yield of 90.58%. The purity was 95.52% by HPLC.erimental Procedure General procedure: In a typical experiment, 0.5mmol of nitroarene and 0.002g(2mol%) NiNPs/DNA were added to 2mL water and thenstirred for 2-3min for thoroughly mixing. Subsequently,1mmol of NaBH4was added to the reaction mixture undermagnetic stirring at room temperature. The extent of thereaction was monitored by thin layer chromatography.Reproducibility of the results was checked by repeating theruns at least three times and was found to be within acceptablelimits (± 3%). When the reaction was completed, thereaction mixture was diluted with ethyl acetate and the catalystwas recovered by centrifugation. The combined organicfractions were dried over Na2SO4and evaporated underreduced pressure. The crude product was purified by columnchromatography on silica gel with a mixture of ethyl acetateand n-hexane as the eluent, and the ratio of ethyl acetate andn-hexane was depended on the structure of the products.The structure of isolated products was verified by 1H NMR. 97%
Safety and Hazards
Pictogram(s)SignalDangerGHS Hazard StatementsH301: Toxic if swallowed H360: May damage fertility or the unborn child H362: May cause harm to breast-fed children H373: Causes damage to organs through prolonged or repeated exposure H401: Toxic to aquatic life H411: Toxic to aquatic life with long lasting effects Precautionary Statement CodesP203, P260, P263, P264, P270, P273, P280, P301+P316, P318, P319, P321, P330, P391, P405, and P501(The corresponding statement to each P-code can be found at the GHS Classification page.)
Other Data
TransportationUnder the room temperature and away from lightHS CodeStorageUnder the room temperature and away from lightShelf Life5 yearsMarket Price
DruglikenessLipinski rules componentMolecular Weight290.322logP0.783HBA4HBD2Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)105.51Rotatable Bond (RotB)5Matching Veber Rules2
Use PatternTrimethoprim CAS#: 738-70-5 is a lipophilic weakly basic pyrimethamine bacteriostatic agent.
https://www.chemwhat.com/trimethoprim%ef%bc%9btrimethoprimcrystalline%ef%bc%88tmp%ef%bc%89-cas-738-70-5/
Comments
Post a Comment