IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
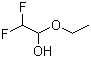
Product Name1-Ethoxy-2,2-Difluoroethan-1-olIUPAC Name1-ethoxy-2,2-difluoroethanol Molecular StructureCAS Registry Number 148992-43-2EINECS NumberMDL NumberMFCD01321160Beilstein Registry NumberSynonyms1-Ethoxy-2,2-difluoroethanol148992-43-2Difluoroacetaldehyde Ethyl Hemiacetal1-ethoxy-2,2-difluoroethan-1-olMFCD01321160SCHEMBL6350971-Ethoxy-2,2-difluoro-ethanolDTXSID40374237difluoroacetaldehyde ethylhemiacetaldifluroacetaldehyde ethyl hemiacetalEthanol, 1-ethoxy-2,2-difluoro-BBL103964STL557774AKOS015838744AM803241AS-48264SY007828CS-0179771D2523FT-0691024EN300-64372F30106992D432J-008539Molecular FormulaC4H8F2O2Molecular Weight126.1InChIInChI=1S/C4H8F2O2/c1-2-8-4(7)3(5)6/h3-4,7H,2H2,1H3 InChI KeyWEEOMNFWRCDRJI-UHFFFAOYSA-N Canonical SMILES
Patent InformationPatent IDTitlePublication Date JP2017/128536MANUFACTURING METHOD OF α,α-DIFLUORO ACETALDEHYDE2017CN107438592Preservation method of alpha, alpha-difluoroacetaldehyde alkyl hemiacetal2017JP2016/33114METHOD FOR IMPROVING STORAGE STABILITY OF 2,2-DIFLUOROACETALDEHYDE2016US2013/79324PYRROLOPYRIMIDINE AND PURINE DERIVATIVES2013
Physical Data
AppearanceColorless to light yellow liquid
Melting Point, °C Solvent (Melting Point) No data available
Boiling Point, °C45 - 47
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzChemical shifts, Spectrum1Hchloroform-d1400Chemical shifts, Spectrum13Cchloroform-d1150Chemical shifts1Hchloroform-d1
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Temperature (IR Spectroscopy), °CBandsKBr
Route of Synthesis (ROS)
Route of Synthesis (ROS) of 1-ethoxy-22-difluoroethan-1-ol-cas-148992-43-2
ConditionsYieldWith lithium aluminium tetrahydride In tetrahydrofuran; diethyl ether at -78℃; for 2.75h;100%With lithium aluminium tetrahydride In tetrahydrofuran at -78℃; for 4.5h; Time; Inert atmosphere;84%With hydrogen In 2-methyltetrahydrofuran; water at 40℃; under 15001.5 Torr; for 24h; chemoselective reaction;98%Experimental Procedure Scheme 4, steps A and B:To an oven dried 1 L flask is added ethy1 2,2- difluoroacetate (25.4 mL, 242 mmol) and diethy1 ether (40 mL), and the mixture is cooled to -78 °C. To the mixture is added lithium aluminum hydride (1 M solution in THF, 61.0 mL, 61.0 mmol) dropwise via addition funnel over a period of 25 minutes. The addition funnel is further washed with diethy1 ether (5 mL) and added dropwise. After 2 hours and 45 minutes, the reaction is quenched with the slow addition of EtOH (6 mL), and is allowed to stir at room temperature for 20 minutes. The mixture is then poured into a mixture of concentrated sulfuric acid (15 mL) in crushed ice (200 mL). After stirring for 5 minutes, the mixture is diluted with diethy1 ether (150 mL), poured into a separatory funnel, and the layers are separated. The aqueous layer is extracted once more with diethy1 ether (200 mL), and the combined organics are dried over magnesium sulfate, filtered, and concentrated in vacuo to give the intermediate 1 -ethoxy-2, 2-difluroethanol in a volume of 45 mL with assumed quantitative yield.erimental Procedure General procedure: In a typical experiment, 0.5mmol of nitroarene and 0.002g(2mol%) NiNPs/DNA were added to 2mL water and thenstirred for 2-3min for thoroughly mixing. Subsequently,1mmol of NaBH4was added to the reaction mixture undermagnetic stirring at room temperature. The extent of thereaction was monitored by thin layer chromatography.Reproducibility of the results was checked by repeating theruns at least three times and was found to be within acceptablelimits (± 3%). When the reaction was completed, thereaction mixture was diluted with ethyl acetate and the catalystwas recovered by centrifugation. The combined organicfractions were dried over Na2SO4and evaporated underreduced pressure. The crude product was purified by columnchromatography on silica gel with a mixture of ethyl acetateand n-hexane as the eluent, and the ratio of ethyl acetate andn-hexane was depended on the structure of the products.The structure of isolated products was verified by 1H NMR.97%
Safety and Hazards
Pictogram(s)SignalWarningGHS Hazard StatementsH226 (100%): Flammable liquid and vapor H315 (18.18%): Causes skin irritation H319 (18.18%): Causes serious eye irritation H335 (27.27%): May cause respiratory irritation Precautionary Statement CodesP210, P233, P240, P241, P242, P243, P261, P264, P264+P265, P271, P280, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P370+P378, P403+P233, P403+P235, P405, and P501(The corresponding statement to each P-code can be found at the GHS Classification page.)
Other Data
TransportationUnder the room temperature and away from lightHS CodeStorageUnder the room temperature and away from lightShelf LifeHalf of a yearMarket Price
DruglikenessLipinski rules componentMolecular Weight126.103logP0.817HBA2HBD1Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)29.46Rotatable Bond (RotB)3Matching Veber Rules2
Quantitative ResultsNo data available
Use Pattern1-Ethoxy-2,2-Difluoroethan-1-ol CAS#: 148992-43-2 is used in pharmaceutical intermediates.
https://www.chemwhat.com/1-ethoxy-22-difluoroethan-1-ol-cas-148992-43-2/
Comments
Post a Comment