IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
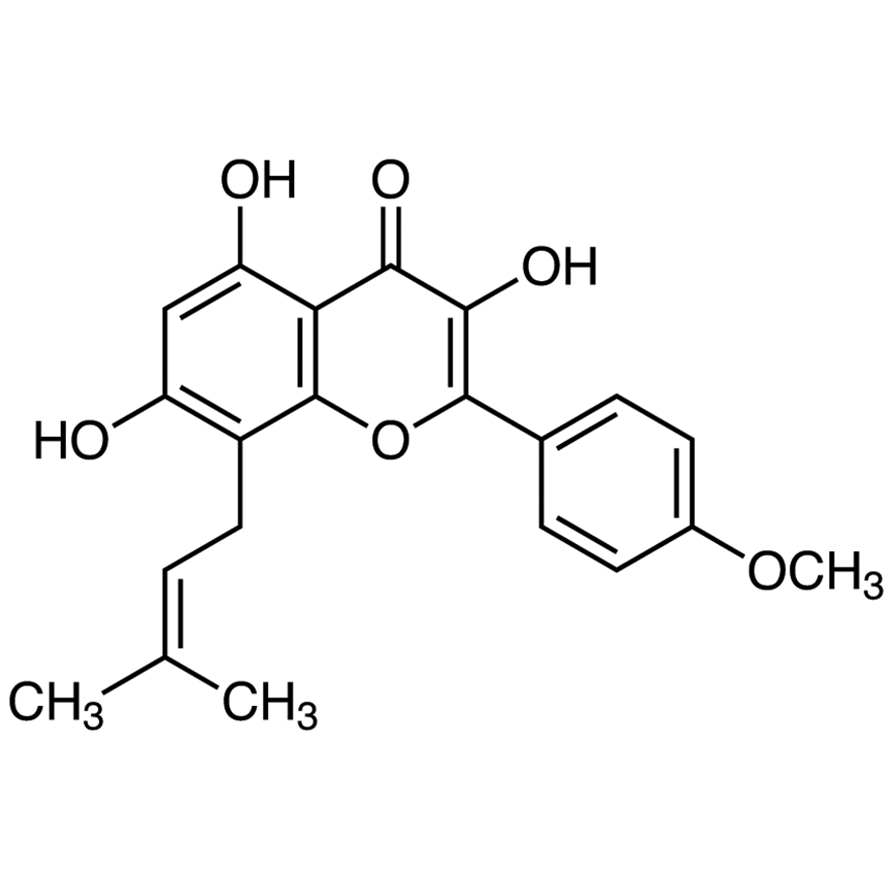
Product NameIcariinIUPAC Name3,5,7-trihydroxy-2-(4-methoxyphenyl)-8-(3-methylbut-2-enyl)chromen-4-one Molecular Structure
 CAS Registry Number 118525-40-9EINECS NumberNo available dataMDL NumberMFCD22422519Beilstein Registry NumberNo available dataSynonymsicaritinanhydroicaritin3,5,7-trihydroxy-2-(4-methoxyphenyl)-8-(3-methylbut-2-en-1-yl)-4H-chromen-4-oneicariinAHIICTMolecular FormulaC21H20O6Molecular Weight368.386InChIInChI=1S/C21H20O6/c1-11(2)4-9-14-15(22)10-16(23)17-18(24)19(25)20(27-21(14)17)12-5-7-13(26-3)8-6-12/h4-8,10,22-23,25H,9H2,1-3H3 InChI KeyTUUXBSASAQJECY-UHFFFAOYSA-N Canonical SMILESCC(=CCC1=C2C(=C(C=C1O)O)C(=O)C(=C(O2)C3=CC=C(C=C3)OC)O)C
CAS Registry Number 118525-40-9EINECS NumberNo available dataMDL NumberMFCD22422519Beilstein Registry NumberNo available dataSynonymsicaritinanhydroicaritin3,5,7-trihydroxy-2-(4-methoxyphenyl)-8-(3-methylbut-2-en-1-yl)-4H-chromen-4-oneicariinAHIICTMolecular FormulaC21H20O6Molecular Weight368.386InChIInChI=1S/C21H20O6/c1-11(2)4-9-14-15(22)10-16(23)17-18(24)19(25)20(27-21(14)17)12-5-7-13(26-3)8-6-12/h4-8,10,22-23,25H,9H2,1-3H3 InChI KeyTUUXBSASAQJECY-UHFFFAOYSA-N Canonical SMILESCC(=CCC1=C2C(=C(C=C1O)O)C(=O)C(=C(O2)C3=CC=C(C=C3)OC)O)C Patent InformationPatent IDTitlePublication DateWO2022/104153COMPOUNDS AND METHODS FOR TREATING VIRAL INFECTIONS 2022CN113180043Application of chitin hydrolase inhibitor in regulation of insect growth activity2021CN113024602Phosphate ester derivative of herba epimedii as well as preparation method and application of phosphate ester derivative2021
Physical Data
AppearanceYellow needle crystal or yellow crystalline powderSolubilityNo data availableFlash PointNo data availableRefractive indexNo data availableSensitivityNo data available
Melting Point, °C Solvent (Melting Point) 178 - 180253207 - 208methanol231 - 232methanol
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Frequency (NMR Spectroscopy), MHzChemical shifts, Spectrum1H
dimethylsulfoxide-d6400HMBC (Heteronuclear Multiple Bond Coherence), Spectrum1H, 13Cdimethylsulfoxide-d6No data available
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Temperature (IR Spectroscopy), °CBandspotassium bromide 27Bandspotassium bromide14.85 - 54.85
Description (UV/VIS Spectroscopy)Solvent (UV/VIS Spectroscopy)Comment (UV/VIS Spectroscopy)Absorption Maxima (UV/VIS), nmExt./Abs. Coefficient, l·mol-1cm-1SpectrummethanolNo data available271, 321, 372No available dataAbsorption maxima
methanolNo data available
368No available data
Route of Synthesis (ROS)
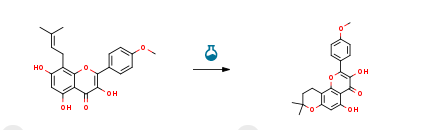 Route of Synthesis (ROS) of Icariin CAS #: 118525-40-9
Route of Synthesis (ROS) of Icariin CAS #: 118525-40-9ConditionsYieldWith sulfuric acid; glacial acetic acid at 90℃; for 6h;92%With hydrogen; triethylamine In ethanol; water at 110℃; under 30003 Torr; for 24h; Autoclave;98%
Safety and Hazards
GHS Hazard StatementsNot Classified
Other Data
TransportationStore at room temperature, sealed and away from lightHS CodeNo data availableStorageStore at 2-8℃ for long time, Sealed and away from lightShelf Life1 year
DruglikenessLipinski rules componentMolecular Weight368.386logP4.751HBA2HBD3Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)96.22Rotatable Bond (RotB)4Matching Veber Rules2
BioactivityPharmacological Data
1 of 3Comment (Pharmacological Data)Bioactivities presentReferenceCurrent Patent Assignee: AMOREPACIFIC CORP. - WO2007/64085, 2007, A12 of 3Comment (Pharmacological Data)Bioactivities presentReferenceCurrent Patent Assignee: BEIJING SHENOGEN PHARMA GROUP - WO2008/52005, 2008, A23 of 3Comment (Pharmacological Data)Bioactivities presentReferenceCurrent Patent Assignee: LUNAN PHARMACEUTICAL GROUP CO LTD - US2016/250243, 2016, A1
Use PatternIcariin CAS #:118525-40-9 is used Pharmaceuticals and prevent thrombocytopenia,preventing bone marrow suppression
https://www.chemwhat.com/icariin-cas-118525-40-9/
Comments
Post a Comment