
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
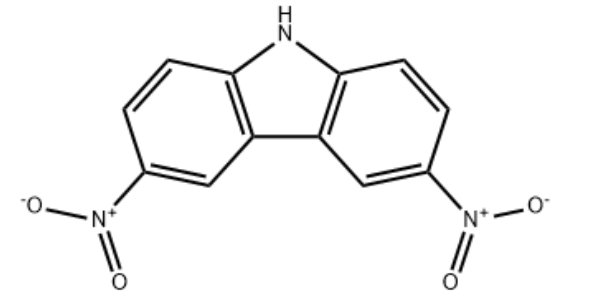
Product Name3,6-Dinitro-9H-carbazoleIUPAC Name3,6-dinitro-9H-carbazole Molecular StructureCAS Registry Number 3244-54-0EINECS Number221-814-4Synonyms3-Pyridinamin;3-Pyridinamine;3-Pyridinamine;pyridin-3-amine;T6NJ CZ;3- Aminopyridine;3-Amino-pyridine;3-pyridylamine;Amino-3 pyridine;m-Aminopyridine;MS/MS-1064463;Pyridin-3-y3,6-Dinitro-9H-carbazole3244-54-03,6-Dinitrocarbazole9H-Carbazole, 3,6-dinitro-L5YYP3DVN99H-Carbazole,3,6-dinitro-Carbazole, 3,6-dinitro-CCRIS 6772EINECS 221-814-4Maybridge1_002104UNII-L5YYP3DVN9Oprea1_673803SCHEMBL155013SCHEMBL631957HMS547H14DTXSID80186179STK673606AKOS003653972AS-75794ST4032785CS-0094238NS00029280N1691310.14272/IARXLBTXILYASE-UHFFFAOYSA-N.1EN300-18547434doi:10.14272/IARXLBTXILYASE-UHFFFAOYSA-N.1Q63408793InChI=1/C12H7N3O4/c16-14(17)7-1-3-11-9(5-7)10-6-8(15(18)19)2-4-12(10)13-11/h1-6,13Molecular FormulaC12H7N3O4Molecular Weight257.2InChIInChI=1S/C12H7N3O4/c16-14(17)7-1-3-11-9(5-7)10-6-8(15(18)19)2-4-12(10)13-11/h1-6,13HInChI KeyIARXLBTXILYASE-UHFFFAOYSA-NSMILESC1=CC2=C(C=C1(=O))C3=C(N2)C=CC(=C3)(=O)
Patent InformationPatent IDTitlePublication DateCN113200906Carbazole triamine derivative and preparation method and application thereof2021US2004/138301Chemical uncouplers for the treatment of obesity2004
Physical Data
Melting Point, °C Solvent (Melting Point) 242 - 244242 - 243242 - 243300383 - 385dimethylformamide351386 - 387nitrobenzene
Description (Association (MCS))Solvent (Association (MCS))Temperature (Association (MCS)), °CPartner (Association (MCS))Stability constant of the complex with ...acetonitrile26.853,6-diamino-9H-carbazole
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Frequency (NMR Spectroscopy), MHzChemical shifts1Hdimethylsulfoxide-d6Chemical shifts1Hdimethylsulfoxide-d6400Chemical shifts1Hacetonitrile400Chemical shifts, Spectrum1Hdimethylsulfoxide-d6400Chemical shifts1Hdimethylsulfoxide-d6400Chemical shifts1Hdimethylsulfoxide-d6400Spectrum1Hdimethylsulfoxide-d6300.151
3,6-Dinitro-9H-carbazole CAS#: 3244-54-0 NMR
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Bandspotassium bromideBandspotassium bromideBandspotassium bromideSpectrumKBrBandsKBr
Description (UV/VIS Spectroscopy)Solvent (UV/VIS Spectroscopy)Comment (UV/VIS Spectroscopy)Absorption Maxima (UV/VIS), nmExt./Abs. Coefficient, l·mol-1cm-1acetonitrileRemark: 298 K260, 320, 360100000, 90000, 47344SpectrumacetonitrileRemark: 298 KUV excited state absorptionSpectrumacetonitrileUV/VISSpectrumSpectrumaq. 1,2-diamino-ethane350 - 550 nm
Route of Synthesis (ROS)
Route of Synthesis (ROS) of 3,6-Dinitro-9H-carbazole CAS# 3244-54-0
ConditionsYieldWith copper(II) nitrate monohydrate; acetic anhydride; acetic acid at 100℃; for 0.5h;Experimental Procedure3.1 2.3.1. Synthesis of 3,6-Dinitrocarbazole (1)Cu(NO3)2·H2O (5.23 g, 22 mmol) was added into a mixture of acetic acid (10 mL) and acetic anhydride (25 mL) at room temperature. The mixture was stirred for 10-20 min, and to this solution was then added carbazole (3.00 g, 18 mmol) slowly in portions over 5 min . Heat was generated during the addition (temperature rose to around 100 °C), and an additional 10 mL of acetic acid was added. The mixture was stirred at this temperature for 30 min and then poured into distilled water (200 mL). The precipitate was collected by filtration, washed with water (100 mL × 3). The still wet product was then dissolved into a potassium alcoholic aqueous solution (KOH 25 g, water 250 mL, and ethanol 250 mL). The solution turned red. After being stirred for 30 min, the solution was filtrated and the filtrate was acidified with concentrated hydrochloric acid. The yellow precipitate was then collected by filtration, washed with water and dried at 100 °C under vacuum to give yellow solid 3.94 g (85%). Mp: 242-243 °C; FT-IR (KBr, cm-1): 3091 (N-H stretching), 1508, 1328 (-NO2 asymmetric and symmetric stretching), 860 (C-N); 1H NMR (400 MHz, DMSO-d6) δ 11.10 (s, 1 H, NH), 9.34 (d, 2 H, J = 2.3 Hz), 8.40 (dd, 2 H, J = 2.3 Hz, J = 8.9 Hz), 7.78 (d, 2 H, J = 8.9 Hz).85%With copper(II) nitrate trihydrate; acetic anhydride; acetic acid at 100℃; for 0.5h;Experimental Procedure1.1 Synthesis of (1) 3, 6-dinitro-carbazoleA mixture of Cu (N03) 2 · 3H20 (5. 23g, 22. OOmmol) was added to a 100mL three-necked flask, and thenWere added 10mL of acetic acid and 20mL of acetic anhydride, stirred at room temperature for 10min to dissolve the solid; weighed carbazole (3. 00g,18. OOmmol) batch was slowly added to the solution to produce a lot of heat during the addition, the temperature rose to about 100 ° C, supplementedAdd 10mL of acetic acid, the reaction 30min. Completion of the reaction, the reaction mixture was poured into 200mL of distilled water, stirred, filtered, washed with distilled waterWashed until the eluate is neutral; filtration cake obtained is dissolved in a first alkali (manufactured by 25g K0H, 250mL of distilled water, 250mLDubbed ethanol), stirred for 30min, filtered to give a brown filtrate with concentrated hydrochloric acid (36% mass fraction of concentrated hydrochloric acid) was acidified filtrateTo a large number of yellow solid was precipitated, the precipitate was filtered, washed with water several times, and dried to give a yellow solid 3. 94g, that is, 3, 6-Nitro carbazole, 85% yield.85%With copper nitrate hemi(pentahydrate); acetic anhydride; acetic acid at 15 - 90℃; for 1.16667h;Experimental Procedure4.1.1. 3,6-Dinitro-9H-carbazole (2)A homogeneous mixture of Cu(NO3)2·2.5H2O (30 mmol), acetic acid (20 mL), and acetic anhydride (30 mL) was prepared at room temperature. To this solution were added carbazole (25 mmol) in small portions over 10 min. Temperature was maintained at 15-20 °C during addition of carbazole. The temperature was allowed to rise to room temperature over a period of 30 min and then to 90 °C. Reaction was continued with stirring for a period of 30 min at this temperature. The mixture was poured into 250 mL of distilled water with constant stirring. The precipitate was collected by filtration, and washed five times each with about 100 mL of distilled water. The filtrate was dried in vacuumat. Chromatography on silica gel, eluting with petroleum/EtOAC (3:1) gave 2 (5.23 g, 81%) as a yellow solid. mp >300 °C (lit.27 mp >300 °C). 1H NMR (400 MHz, DMSO-d6) δ 12.69 (s, 1H), 9.50 (d, J = 2.1 Hz, 2H), 8.40 (dd, J = 9.0, 2.1 Hz, 2H), 7.77 (d, J = 9.0 Hz, 2H). ESI-MS m/z: 258+.81%
Safety and Hazards
No data available
Other Data
TransportationUnder room temperature away from lightHS CodeStorageUnder room temperature away from lightShelf Life1 yearMarket Price
DruglikenessLipinski rules componentMolecular Weight257.205logP3.315HBA1HBD1Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)107.43Rotatable Bond (RotB)2Matching Veber Rules2
Use Pattern3,6-Dinitro-9H-carbazole is a compound with fluorescence properties and can be used in the preparation of luminescent materials. It may have potential optoelectronic performance in organic light-emitting diodes (OLEDs) and fluorescent dyes. And Due to its unique color and light-absorbing properties, 3,6-dinitro-9H-carbazole can be used as a component in pigments for dyes, paints, and inks.It's important to note that the preparation and use of explosives are illegal and dangerous. However, as 3,6-dinitro-9H-carbazole is a nitro-substituted compound, its nitro functional groups may give it explosive properties. Nonetheless, the production and use of explosives are highly regulated and require specialized permits and knowledge. https://www.chemwhat.com/36-dinitro-9h-carbazole-cas-3244-54-0/
Comments
Post a Comment