
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
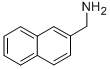
Product Name1-(2-NAPHTHYL)METHANAMINEIUPAC Namenaphthalen-2-ylmethanamineMolecular StructureCAS Registry Number 2018-90-8MDL NumberMFCD01529867Synonyms1-(2-NAPHTHYL)METHANAMINE899-666-02018-90-82-Naphthalenemethanaminenaphthalen-2-ylmethanamine2-(Aminomethyl)naphthalene2-naphthylmethanamineMFCD015298672-Aminomethylnaphthalene(naphthalen-2-yl)methanamine(naphthalene-2-yl)methylamineNaphthalen-2-methylamineC-Naphthalen-2-yl-methylamine2-naphthylmethylamine2-Aminomethylnapthalene2-naphthalenemethylamineSCHEMBL42223SCHEMBL131652SCHEMBL2003901SCHEMBL6643495SCHEMBL6897074C-naphethalene-2-yl-methylamineSCHEMBL27678365(C-naphthalene-2-yl)methylamineDTXSID40174012XBCAHQUVHHVHHL-UHFFFAOYSA-NAKOS000133202CS-W002778FN15510SB76533BS-13623SY023509EN300-33147AB01018358-011-(2-Naphthyl) methanamine;Naphthalen-2-methylamineMolecular FormulaC11H11N Molecular Weight157.21InChIInChI=1S/C11H11N/c12-8-9-5-6-10-3-1-2-4-11(10)7-9/h1-7H,8,12H2InChI KeyXBCAHQUVHHVHHL-UHFFFAOYSA-NSMILESC1=CC=C2C=C(C=CC2=C1)CN
Patent InformationPatent IDTitlePublication DateCN114805106Preparation method of amide compound2022CN114773252Chiral amino indoline derivative as well as preparation method and application thereof2022CN106928037Preparation method of carvone2017CN107417623One-step synthesis method of 5-diarylamino benzimidazole derivatives2017EP2810944HETEROCYCLIC COMPOUND HAVING ANTI-HIV ACTIVITY2014US2007/179115Purinenucleoside derivative modified in 8-position and medical use thereof2007US6337398Succinoylamino hydroxyethylamino sulfonyl urea derivatives useful as retroviral protease inhibitors2002
Physical Data
AppearanceWhite powder
Melting Point, °C 55 - 5755 - 5655 - 562695859 - 6060
Boiling Point, °CPressure (Boiling Point), Torr143 - 147818024148 - 14912
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Frequency (NMR Spectroscopy), MHzChemical shifts, Spectrum1Hdimethylsulfoxide-d6Chemical shifts, Spectrum13Cdimethylsulfoxide-d6Chemical shifts, Spectrum1Hchloroform-d1Chemical shifts1Hchloroform-d1Chemical shifts, Spectrum1Hchloroform-d1400.1Chemical shifts, Spectrum13Cchloroform-d1100.6Chemical shifts, Spectrum1Hchloroform-d1
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Bandspotassium bromideATR (attenuated total reflectance), Bandsneat (no solvent, solid phase)ATR (attenuated total reflectance), BandsFT-IR, in KBrBandsKBrBandsKBr
Route of Synthesis (ROS)
Route of Synthesis (ROS) of 1-(2-NAPHTHYL)METHANAMINE CAS 2018-90-8
ConditionsYieldWith piperidin-2-one; sodium tetrahydroborate; N,N-diisopropyl-4H-benzodioxaborinin-2-amine; ammonia In 1,2-dichloro-ethane at 20℃;99%With ammonia; hydrogen In methanol at 120℃; under 15001.5 Torr; for 4h; Autoclave;Experimental ProcedureA method to catalyze the reductive amination of aldehydes and ketones to prepare primary amines, except that “benzaldehyde” in step 2) of Example 1 is replaced with “2-naphthaldehyde” and “reaction at 90°C for 4h” is replaced with “120°C” Except for the reaction for 4h", everything else was exactly the same as in Example 1. The yield of 2-naphthylmethylamine was 99%.99%With ammonium hydroxide; hydrogen In ethanol at 130℃; under 7500.75 Torr; for 12h; Autoclave;Experimental Procedure2.4. General procedure of the reductive aminationGeneral procedure: The reductive amination of carbonyl compounds was performed in a 50 mL stainless steel autoclave reactor. In a typical run, benzaldehyde (1 mmol), Co(at)NC-800 (20 mg), ethanol (8 mL) and NH3.H2O (26.5 wt%, 2 mL) were charged into the reactor, and then the autoclave reactor was closed. The reactor was flushed with H2 for several times to remove air, and then charged with 1 MPa H2 at room temperature. The reaction was then carriedout at 130 °C for 12 h with a stirring rate of 1000 RPM. After reaction,the reaction mixture was cooled down to room temperature and then depressurized. Then, the products in the reaction mixture were detected by gas chromatography by the use of ethylbenzene as the internal standard. The products were also identified by GC/MS (Shimadzu GCMS-QP2010) equipped with Agilent capillary column DB-5MS.93.6%
Safety and Hazards
Pictogram(s)SignalWarningGHS Hazard StatementsH302 (50%): Harmful if swallowed H315 (100%): Causes skin irritation H319 (100%): Causes serious eye irritation H335 (100%): May cause respiratory irritation H411 (50%): Toxic to aquatic life with long lasting effects Precautionary Statement CodesP261, P264, P264+P265, P270, P271, P273, P280, P301+P317, P302+P352, P304+P340, P305+P351+P338, P319, P321, P330, P332+P317, P337+P317, P362+P364, P391, P403+P233, P405, and P501(The corresponding statement to each P-code can be found at the GHS Classification page.)
Other Data
TransportationStore at around 0 ℃HS CodeStorageStore at around 0 ℃Shelf Life1 yearMarket Price
DruglikenessLipinski rules componentMolecular Weight157.215logP2.41HBA1HBD1Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)26.02Rotatable Bond (RotB)1Matching Veber Rules2
Use Pattern2-(Aminomethyl)naphthalene CAS 2018-90-8 is mainly used as an intermediate in pharmaceutical synthesis and organic chemistry for producing functional compounds. https://www.chemwhat.com/1-2-naphthylmethanamine-cas-2018-90-8/
Comments
Post a Comment