
IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
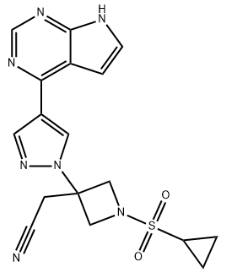
Product NameIlunocitinibIUPAC Name2-pyrimidin-4-yl)pyrazol-1-yl]azetidin-3-yl]acetonitrileMolecular StructureCAS Registry Number 1187594-14-4SynonymsIlunocitinib1187594-14-4Ilunocitinib N3TB5AH8B9LY3411067LY-34110672-pyrimidin-4-yl)pyrazol-1-yl]azetidin-3-yl]acetonitrileILUNOCITINIB UNII-N3TB5AH8B9WHO 118943-Azetidineacetonitrile, 1-(cyclopropylsulfonyl)-3-(4-(7H-pyrrolo(2,3-d)pyrimidin-4-yl)-1H-pyrazol-1-yl)-(1-(Cyclopropanesulfonyl)-3-(4-(7H-pyrrolo(2,3-d)pyrimidin-4-yl)-1H-pyrazol-1-yl)azetidin-3-yl)acetonitrile1-(CYCLOPROPYLSULFONYL)-3-(4-(7H-PYRROLO(2,3-D)PYRIMIDIN-4-YL)-1H-PYRAZOL-1-YL)-3-AZETIDINEACETONITRILE2-(1-cyclopropylsulfonyl-3-(4-(7H-pyrrolo(2,3-d)pyrimidin-4-yl)pyrazol-1-yl)azetidin-3-yl)acetonitrileSCHEMBL870650CHEMBL4802241GTPL11670RVOUEXFKIYNODQ-UHFFFAOYSA-NEX-A9647AKOS040756527DA-74442MS-26294HY-132819CS-0204093G185072-(3-(4-(7h-pyrrolopyrimidin-4-yl)-1h-pyrazol-1-yl)-1-(cyclopropylsulfonyl)azetidin-3-yl)acetonitrileMolecular FormulaC17H17N7O2SMolecular Weight383.4InChIInChI=1S/C17H17N7O2S/c18-5-4-17(9-23(10-17)27(25,26)13-1-2-13)24-8-12(7-22-24)15-14-3-6-19-16(14)21-11-20-15/h3,6-8,11,13H,1-2,4,9-10H2,(H,19,20,21)InChI KeyRVOUEXFKIYNODQ-UHFFFAOYSA-NIsomeric SMILESC1CC1S(=O)(=O)N2CC(C2)(CC#N)N3C=C(C=N3)C4=C5C=CNC5=NC=N4
Patent InformationPatent IDTitlePublication DateUS2009/233903AZETIDINE AND CYCLOBUTANE DERIVATIVES AS JAK INHIBITORS2009
Physical Data
AppearanceWhite powder
Spectra
Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Frequency (NMR Spectroscopy), MHz1Hdimethylsulfoxide-d6400
Route of Synthesis (ROS)
Route of Synthesis (ROS) of Ilunocitinib CAS 1187594-14-4
ConditionsYieldWith phosphoric acid In ethanol; acetonitrile at 20 - 67℃;Experimental Procedure81 2-(3-(4-(7H-Pyrrolopyrimidin-4-yl)-1H-pyrazol-1-yl)-1-(cyclopropylsulfonyl)azetidin-3-yl)acetonitrile phosphoric acid saltTo a 1 L round bottom flask equipped with a stir bar, an addition funnel, a nitrogen inlet and a condenser was charged 2-(3-(4-(7H-pyrrolopyrimidin-4-yl)-1H-pyrazol-1-yl)-1-(cyclopropylsulfonyl)azetidin-3-yl)acetonitrile (27, 11.3 g, 29.5 mmol) and acetonitrile (275 ML) at room temperature. The resulting mixture was warmed to 70° C. before being charged with ethanol (EtOH, 150 mL) in three portions at 70° C. The resulting homogeneous solution was filtered into a clean 1 L round bottom flask equipped with the overhead stirring, an addition funnel, a nitrogen inlet and a condenser. The mixture was then warmed to 67° C. producing a homogeneous solution again. A solution of phosphoric acid (H3PO4, 3.03 g, 30.9 mmol, 1.05 equiv) in ethanol (EtOH, 30 mL) was then charged drop wise to the solution over ten minutes at 67° C. The solution was still homogeneous after the end of the addition of phosphoric acid ethanol solution. The resulting reaction mixture was stirred at 67° C. for 10 minutes before being gradually cooled down to room temperature and stirred at room temperature for 19 h. The solids were collected by filtration and washed with acetonitrile (2×40 mL). The wet cake was partially dried under high vacuum and then transferred to a 75° C. vacuum oven and dried to constant weight to afford 2-(3-(4-(7H-pyrrolopyrimidin-4-yl)-1H-pyrazol-1-yl)-1-(cyclopropylsulfonyl)azetidin-3-yl)acetonitrile phosphate (12.0 g, 14.2 g theoretical, 84.5% yield) as white solids. For Phosphate: 1H NMR (DMSO-d6, 500 MHz) δ 12.13 (br. s, 1H), 9.20 (br. s, 3H), 8.94 (s, 1H), 8.70 (s, 1H), 8.47 (s, 1H), 7.61 (dd, 1H, J=3.4, 2.3 Hz), 7.07 (dd, 1H, J=3.6, 1.6 Hz), 4.65 (d, 2H, J=9.1 Hz), 4.28 (d, 2H, J=9.7 Hz), 3.68 (s, 2H), 2.82 (m, 1H), 1.01 (m, 4H) ppm; 13C NMR (DMSO-d6, 125 MHz) δ 152.9, 151.6, 150.0, 140.6, 130.3, 127.7, 122.9, 117.3, 113.8, 100.7, 59.7, 57.1, 27.6, 25.4, 4.9 ppm; C17H20N7O6PS (MW, 481.42; C17H17N7O2S for free base, MW, 383.43), LCMS (EI) m/e 384 (M++H).84.5%
Safety and Hazards
GHS Hazard StatementsNot Classified
Source: European Chemicals Agency (ECHA)License Note: Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: “Source: European Chemicals Agency, http://echa.europa.eu/”. Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page.License URL: https://echa.europa.eu/web/guest/legal-noticeRecord Name: (1-Cyano-2-ethoxy-2-oxoethylidenaminooxy)dimethylamino-morpholino-carbenium hexafluorophosphateURL: https://echa.europa.eu/information-on-chemicals/cl-inventory-database/-/discli/details/213446Description: The information provided here is aggregated from the “Notified classification and labelling” from ECHA’s C&L Inventory. Read more: https://echa.europa.eu/information-on-chemicals/cl-inventory-database
Other Data
TransportationStore at 2~8°C, away from light.HS CodeStorageStore at 2~8°C, away from light.Shelf Life1 yearMarket Price
DruglikenessLipinski rules componentMolecular Weight383.434logP0.366HBA9HBD1Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)128.94Rotatable Bond (RotB)5Matching Veber Rules2
Use PatternIlunocitinib is a safe, effective, and convenient Janus kinase (JAK) inhibitor specifically designed to manage pruritus (itchiness) associated with allergic dermatitis, as well as to control atopic dermatitis (AD) in dogs aged 12 months and older. As a JAK inhibitor, Ilunocitinib works by interfering with the JAK-STAT signaling pathway, which is critical in mediating inflammation and immune responses in conditions like allergic and atopic dermatitis. By inhibiting this pathway, Ilunocitinib helps to reduce inflammation and alleviate the intense itching that often accompanies these conditions.Atopic dermatitis in dogs is a chronic skin disease that can cause severe discomfort due to persistent itching and inflammation, leading to skin lesions, infections, and a decreased quality of life. Traditional treatments, such as corticosteroids, often come with long-term side effects, making the development of targeted therapies like Ilunocitinib a significant advancement in veterinary medicine.Ilunocitinib offers a more targeted approach to managing the disease by directly inhibiting the pathways involved in the inflammatory response without broadly suppressing the immune system. Its safety profile and ease of administration make it a practical option for long-term management, improving the well-being of affected dogs and offering relief from the burdens of chronic dermatitis. https://www.chemwhat.com/ilunocitinib-cas-1187594-14-4/
Comments
Post a Comment