IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
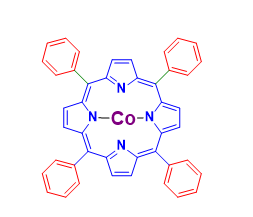
Product NameCobalt tetramethoxyphenylporphyrinIUPAC NameMolecular StructureCAS Registry Number 28903-71-1EINECS NumberNo data availableMDL NumberMFCD00010724Beilstein Registry NumberNo data availableSynonyms5,10,15,20-tetrakis(4-pyridyl)porphyrin zinc(II)zinc-5,10,15,20-tetrakis(N-methylpyridinium-4-yl)porphyrinzinc(II) 5,10,15,20-tetra(4-pyridyl)-21H,23H-porphinezinc 5,10,15,20-tetra(4-pyridinyl)-21H,23H-porphyrin5,10,15,20-tetrakis(4'-pyridyl) zinc(II) porphyrin5,10,15,20-tetrakis(4-pyridinium)porphyrinato zincMolecular FormulaC48H36CoN4O4Molecular Weight791.757InChIInChI=1S/C48H36N4O4.Co/c1-53-33-13-5-29(6-14-33)45-37-21-23-39(49-37)46(30-7-15-34(54-2)16-8-30)41-25-27-43(51-41)48(32-11-19-36(56-4)20-12-32)44-28-26-42(52-44)47(40-24-22-38(45)50-40)31-9-17-35(55-3)18-10-31;/h5-28H,1-4H3;/q-2;+2/b45-37-,45-38-,46-39-,46-41-,47-40-,47-42-,48-43-,48-44-;InChI KeyQBCIMRXPMLWVML-NHZJRHMYSA-NCanonical SMILESCOc1ccc(cc1)/c/2c3/nc(/c(c/4n5/c(c(c6n/c(c(c7n(c2cc7)5)/c8ccc(cc8)OC)/C=C6)/c9ccc(cc9)OC)/cc4)/c1ccc(cc1)OC)C=C3
Patent InformationNo data available
Physical Data
AppearanceDark violet crystalline solidSolubilityNo data availableFlash PointNo data availableRefractive indexNo data availableSensitivityNo data available
Description (Association (MCS))Partner (Association (MCS))Association with compoundvanadium pentoxide xerogel
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzChemical shifts, Spectrum1Hdimethylsulfoxide-d626.84500Chemical shifts1Hdimethylsulfoxide-d6300Spectrum1H25125Spectrum13C25125Spectrum1Hd7-N,N-dimethylformamide
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Temperature (IR Spectroscopy), °CBands, SpectrumIntensity of IR bands, Bands, Spectrumpotassium bromideATR (attenuated total reflectance), Bandspotassium bromide Spectrum25Intensity of IR bands, Bandspotassium bromide
Description (Mass Spectrometry)electrospray ionisation (ESI), spectrumhigh resolution mass spectrometry (HRMS), electrospray ionisation (ESI), spectrum
Description (UV/VIS Spectroscopy)Solvent (UV/VIS Spectroscopy)Absorption Maxima (UV/VIS), nmSpectrumN,N-dimethyl-formamideSpectrumdimethyl sulfoxidein the presence of additive(s), Band assignment, Spectrumwater426Band assignment, Spectrumdimethyl sulfoxide426
Route of Synthesis (ROS)
Route of Synthesis (ROS) of Zinc(II) meso-tetra(4-pyridyl)porphine CAS 31183-11-6
ConditionsYieldIn N,N-dimethyl-formamide at 50℃; for 18h; Inert atmosphere;98%In N,N-dimethyl-formamide at 100℃; for 10h; Schlenk technique; Inert atmosphere;95%In N,N-dimethyl-formamide for 2h; Reflux;84.7%In N,N-dimethyl-formamide at 20℃; for 120h;80%
Safety and Hazards
GHS Hazard StatementsNot ClassifiedFor more detailed information, please visit ECHA C&L website
Other Data
TransportationUnder the room temperature and away from light HS CodeNo data availableStorageUnder the room temperature and away from lightShelf Life2 years Market PriceUSD
DruglikenessLipinski rules componentMolecular Weight682.074HBA8HBD0Matching Lipinski Rules2Veber rules componentPolar Surface Area (PSA)55.92Rotatable Bond (RotB)4Matching Veber Rules2
Use PatternCo(II)-Tetramesitylporphyrin (CoTMPP) is a cobalt-containing porphyrin derivative, significant in the fields of porphyrin chemistry and coordination chemistry. In these compounds, the cobalt atom is centrally located within the porphyrin ring, surrounded by four mesityl groups. This unique structure endows CoTMPP with distinct chemical properties and broad application potential.Porphyrins are organic compounds with highly conjugated systems and stable macrocyclic structures. They can form coordination compounds with various metal ions, leading to widespread applications in optics, electronics, catalysis, and other fields. Due to its unique structure, CoTMPP demonstrates significant value in catalysis, medicine, and materials science.Catalyst: CoTMPP is particularly important in the field of catalysis. It can act as a catalyst for various chemical reactions, including oxidation, reduction, and cyclization reactions. Notably, in organic synthesis, CoTMPP can effectively promote certain reactions, enhancing their selectivity and yield.Biomimetic Materials: Due to its structural similarity to natural porphyrin compounds such as hemoglobin and chlorophyll, CoTMPP is used in the development of biomimetic materials. These materials show potential applications in mimicking photosynthesis and oxygen transport.Sensors: Leveraging its ability to specifically interact with various molecules or ions, CoTMPP has been studied as a chemical sensor for detecting specific chemicals or biomarkers in the environment.Electronic and Optical Materials: Thanks to its excellent electronic and optical properties, CoTMPP is also explored for the preparation of organic semiconductors, organic photovoltaic materials, and organic light-emitting diodes (OLEDs).As a functional material, CoTMPP exhibits broad application prospects in many scientific and industrial fields due to its unique chemical and physical properties. With ongoing research, more applications are expected to be developed, contributing to scientific research and technological advancement.
https://www.chemwhat.com/cobalt-tetramethoxyphenylporphyrin-cas-28903-71-1/
Comments
Post a Comment