IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
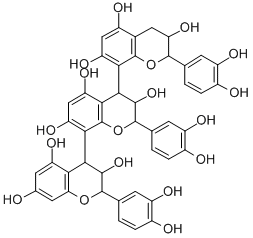
Product NamePROCYANIDIN C1IUPAC Name(2R,3R,4S)-2-(3,4-dihydroxyphenyl)-4--8--3,4-dihydro-2H-chromene-3,5,7-triolMolecular StructureCAS Registry Number 37064-30-5SynonymsProcyanidin C137064-30-5Procyanidol C1Proanthocyanidin C1Cinnamtannin A1Procyanidin trimer C1(2R,2'R,2''R,3R,3'R,3''R,4R,4'S)-2,2',2''-Tris(3,4-dihydroxyphenyl)--3,3',3'',5,5',5'',7,7',7''-nonaolUNII-33516LCW4FCHEBI:7564333516LCW4FEC-(4b,8)-EC-(4b,8)-ECCHEMBL290632(2r,2'r,2''r,3r,3'r,3''r,4r,4's)-2,2',2''-tris(3,4-dihydroxyphenyl)-3,3',3'',4,4',4''-hexahydro-2h,2'h,2''h-4,8':4',8''-terchromene-3,3',3'',5,5',5'',7,7',7''-nonolEpicatechin-(4beta->8)-epicatechin-(4beta->8)-epicatechinPROCYANIDIN C1, (+)-DTXSID101905622-epicatechin(2R,3R,4S)-2-(3,4-dihydroxyphenyl)-4--8--3,4-dihydro-2H-chromene-3,5,7-triol(4,8':4',8''-Ter-2H-1-benzopyran)-3,3',3'',5,5',5'',7,7',7''-nonol, 2,2',2''-tris(3,4-dihydroxyphenyl)-3,3',3'',4,4',4''-hexahydro-, stereoisomer2-epicatechin(Epicatechin(4b->8))2-epicatechinProcyanidin C-1(Epicatechin-(4beta->8))2-epicatechinC45H38O18PCC1SCHEMBL678609DTXCID50113053HY-N2342BDBM50240460AKOS027321198AC-35069MS-316001ST166802CS-0021304C17624Q7247556PROCYANIDIN C1 (CONSTITUENT OF MARITIME PINE)epicatechin-(4beta-8)-epicatechin-(4beta-8)-epicatechinPROCYANIDIN C1 (CONSTITUENT OF MARITIME PINE) Epicatechin-(4.beta.-->8)epicatechin-(4.beta.-->8)epicatechinEPICATECHIN-(4.BETA.->8)-EPICATECHIN-(4.BETA.->8)-EPICATECHIN(2R,3R,4R,2''R,3''R,4''S,2''''R,3''''R)-2,2'',2''''-Tris-(3,4-dihydroxy-phenyl)-3,4,3'',4'',3'''',4''''-hexahydro-2H,2''H,2''''H-terchromene-3,5,7,3'',5'',7'',3'''',5'''',7''''-nonaol(2R,3R,4S)-2-(3,4-dihydroxyphenyl)-4--8-chromane-3,5,7-triol(2R,3R,4S)-2-(3,4-dihydroxyphenyl)-8--4--3,4-dihydro-2H-1-benzopyran-3,5,7-triolMolecular FormulaC45H38O18 Molecular Weight866.8InChIInChI=1S/C45H38O18/c46-18-10-27(54)33-32(11-18)61-42(16-2-5-21(48)25(52)8-16)39(59)37(33)35-29(56)14-30(57)36-38(40(60)43(63-45(35)36)17-3-6-22(49)26(53)9-17)34-28(55)13-23(50)19-12-31(58)41(62-44(19)34)15-1-4-20(47)24(51)7-15/h1-11,13-14,31,37-43,46-60H,12H2/t31-,37-,38+,39-,40-,41-,42-,43-/m1/s1InChI KeyMOJZMWJRUKIQGL-XILRTYJMSA-NIsomeric SMILESC1((OC2=C1C(=CC(=C23((OC4=C(C(=CC(=C34)O)O)5((OC6=CC(=CC(=C56)O)O)C7=CC(=C(C=C7)O)O)O)C8=CC(=C(C=C8)O)O)O)O)O)C9=CC(=C(C=C9)O)O)O
Physical Data
AppearanceBrown powder
Description (Association (MCS))Temperature (Association (MCS)), °CPartner (Association (MCS))Association with compound23.84bovine milk β-lactoglobulinAssociation with compound23.84bovine milk α-lactalbuminAssociation with compound30.84bovine milk β-lactoglobulinAssociation with compound30.84bovine milk α-lactalbuminAssociation with compound37.84bovine milk β-lactoglobulinAssociation with compound37.84bovine milk α-lactalbuminAssociation with compoundbovine serum albumin
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzChemical shifts1HCD3ODChemical shifts13CCD3ODChemical shifts1HCD3OD400Chemical shifts1HCD3OD-18.16400Chemical shifts13CCD3OD-18.16100
Description (IR Spectroscopy)Solvent (IR Spectroscopy)Bandspotassium bromideSpectrumpotassium bromide
Description (UV/VIS Spectroscopy)Absorption Maxima (UV/VIS), nmmethanol200, 280aq. phosphate buffer280water280methanol, water281
Route of Synthesis (ROS)
Route of Synthesis (ROS) of PROCYANIDIN C1 CAS# 37064-30-5
ConditionsYieldWith hydrogen; palladium(II) hydroxide/carbon In tetrahydrofuran; methanol; water under 750.075 Torr; for 1.33333h;Experimental Procedure11.A Example 11; Preparation of Epicatechin (4β,8)-Oligomers from Benzyl-Protected Oligomers; A. Preparation of Epicatechin (4β,8)2-TrimerTo a solution of 64.3 mg (33.0 μmol) of bis(5,7,3',4'-tetra-O-benzyl)epicatechin (4β,8)-dimer in 5 mL of tetrahydrofuran were added 5 mL of methanol, 0.25 mL of water, and 57 mg of 20% Pearlman's catalyst (Pd(OH)2/C). The mixture was stirred under 1 bar of hydrogen for 80 minutes and filtered over cotton. The filtration residue was washed two times with 10 mL of methanol. The combined filtrates were evaporated, and the residue was taken up in 10 mL of HPLC grade water. The solution was filtered and lyophilized to yield 32.4 mg (101%) of epicatechin (4β,8)2-trimer 6H2O as a fluffy, amorphous, off-white solid: D+70.4°, 546+84.40 (MeOH, c 2.2 gL-1); (ref. 4c: D+75.20, acetone, c 8.7 gL-1; ref. 4d: 578+90°, MeOH, c 2 gL-1; ref. 6: D+76.40, acetone, c 8.6 gL-1; ref. 19b: 578+920, H2O, c 1.9 gL-1; ref. 19k: D+800, MeOH, c 1.6 gL-1); 13C NMR (CD3OD, TMS; δ 60-85 region only) δ 79.73, 77.08, 73.47, 72.94, 66.84; MS (API/ES) m/z 865.4 (100%; calcd for -: 865.2), 577.0 (6%), 288.9 (4%). Analysis: Calculated for C45H38O18.6H2O: C, 55.44; H, 5.17. Found: C, 55.71; H, 5.07.100%With hydrogen; palladium dihydroxide In tetrahydrofuran; methanol95%With hydrogen; palladium dihydroxide In tetrahydrofuran; methanol; water at 20℃; for 1h;95%With 10 wt% Pd(OH)2 on carbon; hydrogen In tetrahydrofuran; methanol; water at 20℃; for 1h;92%
Safety and Hazards
GHS Hazard StatementsNot ClassifiedFor more detailed information, please visit ECHA C&L website
Source: European Chemicals Agency (ECHA)License Note: Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: “Source: European Chemicals Agency, http://echa.europa.eu/”. Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page.License URL: https://echa.europa.eu/web/guest/legal-noticeRecord Name: (1-Cyano-2-ethoxy-2-oxoethylidenaminooxy)dimethylamino-morpholino-carbenium hexafluorophosphateURL: https://echa.europa.eu/information-on-chemicals/cl-inventory-database/-/discli/details/213446Description: The information provided here is aggregated from the “Notified classification and labelling” from ECHA’s C&L Inventory. Read more: https://echa.europa.eu/information-on-chemicals/cl-inventory-database
Other Data
TransportationUnder the room temperature and away from lightHS CodeStorageUnder the room temperature and away from lightShelf Life2 yearsMarket Price
DruglikenessLipinski rules componentMolecular Weight866.786logP3.855HBA3HBD15Matching Lipinski Rules2Veber rules componentPolar Surface Area (PSA)331.14Rotatable Bond (RotB)5Matching Veber Rules1
Use PatternProcyanidin C1 CAS 37064-30-5, a complex polyphenolic compound, falls under the larger category of proanthocyanidins which are widely recognized for their potent antioxidant properties. These substances are found in a variety of plants, including fruits, vegetables, nuts, seeds, and bark, playing crucial roles in plant defense mechanisms against pathogens and environmental stressors. Among the diverse array of proanthocyanidins, Procyanidin C1 is particularly notable for its unique structure and significant health-promoting potentials.Chemically, Procyanidin C1 is a trimer, meaning it consists of three flavonoid units linked together. This structural complexity contributes to its strong antioxidant capacity, surpassing that of simpler flavonoids and even some vitamins known for their antioxidant properties, such as vitamin E and C. The antioxidant activity of Procyanidin C1 is essential in neutralizing free radicals, unstable molecules that can cause oxidative stress leading to cellular damage, aging, and various diseases.The health benefits associated with Procyanidin C1 are vast and have been the subject of numerous scientific studies. Research has shown that it can support cardiovascular health by improving blood circulation, reducing blood pressure, and preventing the oxidation of low-density lipoprotein (LDL) cholesterol, a key factor in the development of atherosclerosis. Moreover, its anti-inflammatory effects are crucial in mitigating chronic inflammation, a root cause of many chronic conditions, including cancer, diabetes, and neurodegenerative diseases.
https://www.chemwhat.com/procyanidin-c1-cas-37064-30-5/
Comments
Post a Comment