IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
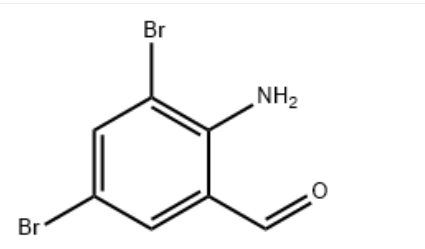
Product Name2-Amino-3,5-dibromobenzaldehydeIUPAC Name2-amino-3,5-dibromobenzaldehyde Molecular StructureCAS Registry Number 50910-55-9EINECS Number256-841-0MDL NumberMFCD00671100Synonyms2-Amino-3,5-dibromobenzaldehyde50910-55-92-amino-3,5-dibromo-benzaldehyde3,5-DIBROMO-2-AMINOBENZALDEHYDE3,5-DibromoanthranilaldehydeBenzaldehyde, 2-amino-3,5-dibromo-C7H5Br2NOMFCD00671100EINECS 256-841-0EC 256-841-0BROMHEXINE IMPURITY BBromhexine EP Impurity BSCHEMBL285148C7-H5-Br2-N-ODTXSID70198943AMY16465BCP06911STL450950Benzaldehido, 2-amino-3,5-dibromo-AKOS0158546962-Amino-3,5-dibromobenzaldehyde, 97%AC-10733AC-31356AS-12042BP-12420SY038949CS-0030731D2625FT-0611038EN300-136846F12175A828361W-105925Ambroxol EP Impurity E; 2-Amino-3,5-dibromobenzaldehydeZ1269136164Molecular FormulaC7H5Br2NOMolecular Weight278.93InChIInChI=1S/C7H5Br2NO/c8-5-1-4(3-11)7(10)6(9)2-5/h1-3H,10H2 InChI KeyRCPAZWISSAVDEA-UHFFFAOYSA-N Canonical SMILESC1=C(C=C(C(=C1C=O)N)Br)Br
Patent InformationPatent IDTitlePublication Date CN1154662122-trifluoromethyl quinoline compound as well as synthesis method and application thereof2022 CN110684031Asymmetric synthesis method of trans-tetrahydropyrine and tetrahydroquinoline derived chiral compound2020CN109096196Preparation method and application of 2-amino-3,5-bibromobenzyl intermediate compound2018
Physical Data
AppearanceYellow Crystalline Powder
Melting Point, °C Solvent (Melting Point) 136.2 - 138.6137 - 139ethanol135 - 136ethanol137 - 137.5
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzChemical shifts, Spectrum1Hdimethylsulfoxide-d6400Chemical shifts, Spectrum13Cdimethylsulfoxide-d6125Chemical shifts, Spectrum1Hchloroform-d1500Chemical shifts, Spectrum13Cchloroform-d1125Chemical shifts, Spectrum1Hchloroform-d1300
Description (IR Spectroscopy)Solvent (IR Spectroscopy)BandsKBrBandsCHCl3
Description (UV/VIS Spectroscopy)Solvent (UV/VIS Spectroscopy)Comment (UV/VIS Spectroscopy)Absorption Maxima (UV/VIS), nmAbsorption maximamethanol234, 261.5
Route of Synthesis (ROS)
Route of Synthesis (ROS) of 2-Amino-3,5-dibromobenzaldehyde CAS 50910-55-9
ConditionsYieldWith bromine; potassium bromide In water91.1%With bromine; potassium bromide In ethanol; water at 20℃; for 1h; Temperature;91.9%With bromine; potassium bromide In ethanol; water for 1h; Ambient temperature;80%Experimental ProcedureStir and mix ethanol and o-aminobenzaldehyde at a molar ratio of 5:1;2. Add a solution of bromine, potassium bromide and water to the reaction solution with stirring, wherein the molar ratio of o-aminobenzaldehyde, bromine and potassium bromide is 1:1.9:9, and the dripping time is 40 minutes;3. Stir the reaction at 20°C for 1 hour and follow the progress of the reaction with high performance liquid chromatography (HPLC);4. Continue adding excess saturated sodium bicarbonate solution to the reaction mixture and stir until solid precipitates and obtain 2-amino-3,5-dibromobenzaldehyde by filtration. The yield of 2-amino-3,5-dibromobenzaldehyde was 91.9%.
Safety and Hazards
No data available
Other Data
TransportationKeep material sealed and store in cool, dry place, away from sunlight and moisture.HS CodeStorageKeep material sealed and store in cool, dry place, away from sunlight and moisture.Shelf Life2 yearsMarket Price
DruglikenessLipinski rules componentMolecular Weight278.931logP2.926HBA2HBD1Matching Lipinski Rules4Veber rules componentPolar Surface Area (PSA)43.09Rotatable Bond (RotB)1Matching Veber Rules2
Toxicity/Safety PharmacologyQuantitative Results
Use Pattern2-Amino-3,5-dibromobenzaldehyde CAS#: 50910-55-9 can serve as an intermediate in organic synthesis. It can participate in various chemical reactions such as amination, electrophilic substitution, condensation reactions, etc., to construct the framework of complex organic molecules or synthesize target compounds.
https://www.chemwhat.com/2-amino-35-dibromobenzaldehyde-cas-50910-55-9-2/
Comments
Post a Comment