IdentificationPhysical DataSpectraRoute of Synthesis (ROS)Safety and HazardsOther Data
Identification
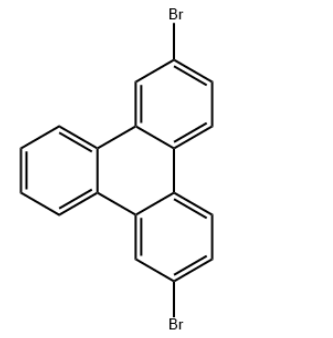
Product Name2,7-DibromotriphenyleneIUPAC Name2,7-dibromotriphenylene Molecular StructureCAS Registry Number 888041-37-0MDL NumberMFCD22571695Synonyms2,7-dibromotriphenylene888041-37-02,7-Dibromo-triphenylene1219091-69-6SCHEMBL12580921DTXSID80583088MFCD22571695AKOS024462942DS-7796CS-0022186D4801FT-0706138F18762A854403Molecular FormulaC18H10Br2Molecular Weight386.1InChIInChI=1S/C18H10Br2/c19-11-5-7-15-16-8-6-12(20)10-18(16)14-4-2-1-3-13(14)17(15)9-11/h1-10HInChI KeyBPGPBYGXGRDFQG-UHFFFAOYSA-NCanonical SMILESC1=CC=C2C(=C1)C3=C(C=CC(=C3)Br)C4=C2C=C(C=C4)Br
Patent InformationPatent IDTitlePublication DateCN108329270Organic electroluminescence material containing triphenylene and benzimidazole structure and organic light-emitting device of organic electroluminescence material2018EP2679581COMPOUND CONTAINING SUBSTITUTED TRIPHENYLE RING STRUCTURE, AND ORGANIC ELECTROLUMINESCENT ELEMENT2014
Physical Data
AppearanceWhite powder
Melting Point, °C Solvent (Melting Point) 220 - 223acetonitrile
Spectra
Description (NMR Spectroscopy)Nucleus (NMR Spectroscopy)Solvents (NMR Spectroscopy)Temperature (NMR Spectroscopy), °C Frequency (NMR Spectroscopy), MHzChemical shifts13CCDCl325125.691Spectrum13CCDCl325125.691Chemical shifts1HCDCl3500Spectrum1HCDCl3500
Description (IR Spectroscopy)Solvent (IR Spectroscopy)BandsNaCl
Route of Synthesis (ROS)
Route of Synthesis (ROS) of 2,7-Dibromotriphenylene CAS 888041-37-0
ConditionsYieldWith palladium bis dichloride; anhydrous potassium acetate In 1,4-dioxane for 72h; Inert atmosphere; Reflux;89%With palladium (II) dichloride; anhydrous potassium acetate In 1,4-dioxane at 85℃; for 12h; Inert atmosphere;76%With dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II) dichloromethane adduct; anhydrous potassium acetate In 1,4-dioxane at 80℃; for 10h; Molecular sieve; Inert atmosphere;70%With dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II) dichloromethane adduct; anhydrous potassium acetate In 1,4-dioxane at 80℃; for 10h; Molecular sieve; Inert atmosphere;70%Experimental Procedure 5.5 g of the obtained 2,7-dibromotriphenylene,7.9 g of bis (pinacolato) diboron,4.2 g of potassium acetate,50 ml of 1,4-dioxane previously dehydrated with Molecular Sieves 4 A,0.4 g of palladium (II) dichloride-dichloromethane complex (1: 1) was added to a reaction vessel purged with nitrogen and heated,Followed by stirring at 80 ° C. for 10 hours.After cooling to 50 ° C.,150 ml of chloroform was added,And the mixture was stirred for 30 minutes.Insoluble matter was removed by filtration,The crude product was obtained by concentration.The crude product was purified by column chromatography ,4.8 g (yield: 70%) of a white powder of 2,7-bis (4,4,5,5-tetramethyl- dioxaborolan-2-yl) triphenylene was obtained.
Safety and Hazards
Pictogram(s)SignalWarningGHS Hazard StatementsH315 (100%): Causes skin irritation H319 (100%): Causes serious eye irritation Precautionary Statement CodesP264, P264+P265, P280, P302+P352, P305+P351+P338, P321, P332+P317, P337+P317, and P362+P364(The corresponding statement to each P-code can be found at the GHS Classification page.)
Other Data
TransportationNot dangerous goodsUnder room temperature away from lightHS CodeStorageUnder room temperature away from lightShelf Life2 yearsMarket Price
DruglikenessLipinski rules componentMolecular Weight386.085logP7.416HBA0HBD0Matching Lipinski Rules3Veber rules componentPolar Surface Area (PSA)0Rotatable Bond (RotB)0Matching Veber Rules2
Toxicity/Safety PharmacologyQuantitative Results
Use Pattern2,7-Dibromotriphenylene CAS 888041-37-0 is uesed as an intermediate in organic synthesis. Through further chemical reactions, it can be used to synthesize organic compounds with different structures and properties. The bromine groups can undergo substitution reactions, allowing for the introduction of other functional groups and expanding the compound's application range.
https://www.chemwhat.com/27-dibromotriphenylene-cas-888041-37-0-2/
Comments
Post a Comment